中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (3): 324-336.doi: 10.12092/j.issn.1009-2501.2026.03.004
张珂1,2( ), 李子婷1,2, 蒋佛佛1,2, 赵锋1, 马银玲1,2,*(
), 李子婷1,2, 蒋佛佛1,2, 赵锋1, 马银玲1,2,*( ), 庞国勋1,2,*(
), 庞国勋1,2,*( )
)
收稿日期:2025-06-11
修回日期:2025-08-07
出版日期:2026-03-26
发布日期:2026-04-03
通讯作者:
马银玲,庞国勋
E-mail:1789884192@qq.com;paper@hebmu.edu.cn;13503291608@163.com
作者简介:张珂,女,硕士研究生,研究方向:药物代谢动力学。E-mail:基金资助:
Ke ZHANG1,2( ), Ziting LI1,2, Fofo JIANG1,2, Feng ZHAO1, Yinling MA1,2,*(
), Ziting LI1,2, Fofo JIANG1,2, Feng ZHAO1, Yinling MA1,2,*( ), Guoxun PANG1,2,*(
), Guoxun PANG1,2,*( )
)
Received:2025-06-11
Revised:2025-08-07
Online:2026-03-26
Published:2026-04-03
Contact:
Yinling MA,Guoxun PANG
E-mail:1789884192@qq.com;paper@hebmu.edu.cn;13503291608@163.com
摘要:
目的: 探究在高脂血症中,丹蒌片(Dan-Lou tablets,DLT)调控阿托伐他汀(atorvastatin,ATV)对大鼠体内药动学及药效学的影响,为异常血脂中西药联用提供理论依据。方法: 采用高脂模型,按临床剂量折算单次/多次灌胃DLT,联合ATV干预后,通过UPLC-MS/MS多离子检测法定量检测大鼠血浆中ATV及其活性代谢物浓度,计算药动参数;同时评估高脂模型血脂指标如总胆固醇(total Cholesterol,TC)、甘油三酯(triglyceride,TG)、高密度脂蛋白胆固醇(high-density lipoprotein cholesterol,HDL-C)和低密度脂蛋白胆固醇(low-density lipoprotein cholesterol,LDL-C),并观察肝组织病理形态的变化。结果: 药动学方面,正常组与模型组中,与单独灌服ATV相比,联合用药后ATV的AUC0-24h分别上升160.63%、50.96%,Cmax分别上升27.95%、7.93%。药效学方面,ATV组与联合给药组分别使高脂大鼠的体质量下降12.48%和29.89%,联合给药组的TC、TG和LDL-C均降低,HDL-C升高,差异具有统计学意义(P<0.01)。肝组织切片显示,联合用药组脂滴分布显著减少,改善效果最为明显。结论: 在高脂异常体质中,DLT能影响ATV在大鼠体内的药动学过程,显著增加ATV的体内暴露量。临床联合用药时,应密切监测疗效及药物不良反应,调整给药方案,避免潜在的药物相互作用风险。
中图分类号:
张珂, 李子婷, 蒋佛佛, 赵锋, 马银玲, 庞国勋. 丹蒌片对阿托伐他汀在高脂饲料喂养大鼠体内药动学与药效学的影响[J]. 中国临床药理学与治疗学, 2026, 31(3): 324-336.
Ke ZHANG, Ziting LI, Fofo JIANG, Feng ZHAO, Yinling MA, Guoxun PANG. Effect of Dan-Lou tablets on the pharmacokinetics and pharmacodynamics of atorvastatin in high-fat diet-fed rats[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(3): 324-336.
| Compound | ATV | o-ATV | p-ATV | CMZ (IS) |
| Quantitative ion transition (m/z) | 559.3→440.6 | 575.3→440.3 | 575.3→440.3 | 237.2→194.2 |
| DP, V | 130 | 91 | 91 | 110 |
| CE, eV | 30 | 33 | 33 | 13 |
表 1
Table 1 MS parameters of the analyte and internal standard
| Compound | ATV | o-ATV | p-ATV | CMZ (IS) |
| Quantitative ion transition (m/z) | 559.3→440.6 | 575.3→440.3 | 575.3→440.3 | 237.2→194.2 |
| DP, V | 130 | 91 | 91 | 110 |
| CE, eV | 30 | 33 | 33 | 13 |
| Group | Body weight (g) | TC (mmol/L) | TG (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) |
| Normal diet group | 185.33±3.96 | 1.39±0.21 | 0.13±0.03 | 2.66±0.11 | 0.46±0.03 |
| High-fat diet group | 373.33±34.58c | 3.36±0.38c | 0.93±0.38c | 0.74±0.11c | 1.03±0.02c |
表 2
Table 2 Comparison of serum lipid profiles between Normal diet and High-fat diet groups in rats (Mean ± SD, n=5)
| Group | Body weight (g) | TC (mmol/L) | TG (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) |
| Normal diet group | 185.33±3.96 | 1.39±0.21 | 0.13±0.03 | 2.66±0.11 | 0.46±0.03 |
| High-fat diet group | 373.33±34.58c | 3.36±0.38c | 0.93±0.38c | 0.74±0.11c | 1.03±0.02c |
| Compound | Concentration (ng/mL) | Intra-day | Inter-day | |||||
| Determined concentrations (ng/mL) | Accuracy (%) | RSD (%) | Determined concentrations (ng/mL) | Accuracy (%) | RSD (%) | |||
| ATV | 0.1 | 0.11±0.005 | 9.00 | 4.93 | 0.11±0.002 | 10.20 | 1.54 | |
| 0.5 | 0.46±0.03 | ?7.49 | 6.15 | 0.47±0.009 | ?6.16 | 2.00 | ||
| 100 | 97.01±3.16 | ?2.99 | 3.26 | 98.24±1.73 | ?1.76 | 1.76 | ||
| 250 | 249.71±4.82 | ?0.11 | 1.93 | 251.43±2.42 | 0.57 | 0.96 | ||
| o-ATV | 0.1 | 0.11±0.003 | 7.58 | 3.14 | 0.11±0.004 | 7.70 | 3.80 | |
| 0.5 | 0.46±0.009 | ?7.19 | 1.93 | 0.46±0.004 | ?7.74 | 0.84 | ||
| 10 | 9.48±0.34 | ?10.50 | 7.52 | 10.54±0.09 | ?4.62 | 1.97 | ||
| 24 | 24.55±0.84 | 2.27 | 3.41 | 24.63±0.12 | 2.61 | 0.47 | ||
| p-ATV | 0.1 | 0.11±0.006 | 12.08 | 5.23 | 0.11±0.008 | 12.72 | 7.07 | |
| 0.5 | 0.52±0.02 | 3.00 | 3.59 | 0.51±0.003 | 2.54 | 0.61 | ||
| 10 | 10.42±0.13 | 4.22 | 1.20 | 10.42±1.39 | 4.19 | 0.13 | ||
| 24 | 22.13±0.66 | ?7.80 | 3.00 | 22.00±0.11 | ?8.34 | 0.52 | ||
表 3
Table 3 Intra-day and inter-day precision and accuracy of ATV, o-ATV and p-ATV in rat plasma by LC-MS/MS (Mean ± SD)
| Compound | Concentration (ng/mL) | Intra-day | Inter-day | |||||
| Determined concentrations (ng/mL) | Accuracy (%) | RSD (%) | Determined concentrations (ng/mL) | Accuracy (%) | RSD (%) | |||
| ATV | 0.1 | 0.11±0.005 | 9.00 | 4.93 | 0.11±0.002 | 10.20 | 1.54 | |
| 0.5 | 0.46±0.03 | ?7.49 | 6.15 | 0.47±0.009 | ?6.16 | 2.00 | ||
| 100 | 97.01±3.16 | ?2.99 | 3.26 | 98.24±1.73 | ?1.76 | 1.76 | ||
| 250 | 249.71±4.82 | ?0.11 | 1.93 | 251.43±2.42 | 0.57 | 0.96 | ||
| o-ATV | 0.1 | 0.11±0.003 | 7.58 | 3.14 | 0.11±0.004 | 7.70 | 3.80 | |
| 0.5 | 0.46±0.009 | ?7.19 | 1.93 | 0.46±0.004 | ?7.74 | 0.84 | ||
| 10 | 9.48±0.34 | ?10.50 | 7.52 | 10.54±0.09 | ?4.62 | 1.97 | ||
| 24 | 24.55±0.84 | 2.27 | 3.41 | 24.63±0.12 | 2.61 | 0.47 | ||
| p-ATV | 0.1 | 0.11±0.006 | 12.08 | 5.23 | 0.11±0.008 | 12.72 | 7.07 | |
| 0.5 | 0.52±0.02 | 3.00 | 3.59 | 0.51±0.003 | 2.54 | 0.61 | ||
| 10 | 10.42±0.13 | 4.22 | 1.20 | 10.42±1.39 | 4.19 | 0.13 | ||
| 24 | 22.13±0.66 | ?7.80 | 3.00 | 22.00±0.11 | ?8.34 | 0.52 | ||
| Compound | Concentration (ng/mL) | Extraction Recovery% | RSD% | Matrix effect% | RSD% |
| ATV | 0.5 | 91.24 | 3.98 | 111.29 | 10.17 |
| 150 | 105.92 | 8.35 | 113.99 | 11.92 | |
| 300 | 103.65 | 11.38 | 109.83 | 12.36 | |
| o-ATV | 0.5 | 97.44 | 7.21 | 97.32 | 5.84 |
| 10 | 92.05 | 7.70 | 102.17 | 11.02 | |
| 24 | 104.63 | 11.64 | 95.12 | 12.89 | |
| p-ATV | 0.5 | 100.14 | 9.12 | 101.22 | 10.98 |
| 10 | 103.35 | 9.86 | 96.32 | 5.94 | |
| 24 | 97.15 | 10.27 | 102.20 | 8.58 |
表 4
Table 4 Matrix effect and extraction recovery of ATV, o-ATV and p-ATV in rat plasma (Mean ± SD)
| Compound | Concentration (ng/mL) | Extraction Recovery% | RSD% | Matrix effect% | RSD% |
| ATV | 0.5 | 91.24 | 3.98 | 111.29 | 10.17 |
| 150 | 105.92 | 8.35 | 113.99 | 11.92 | |
| 300 | 103.65 | 11.38 | 109.83 | 12.36 | |
| o-ATV | 0.5 | 97.44 | 7.21 | 97.32 | 5.84 |
| 10 | 92.05 | 7.70 | 102.17 | 11.02 | |
| 24 | 104.63 | 11.64 | 95.12 | 12.89 | |
| p-ATV | 0.5 | 100.14 | 9.12 | 101.22 | 10.98 |
| 10 | 103.35 | 9.86 | 96.32 | 5.94 | |
| 24 | 97.15 | 10.27 | 102.20 | 8.58 |
| Compound | Concentration (ng/mL) | Room temperature for 2 h | 8 h at 4 ℃ | Three freezes-thraws (?80 ℃) | |||||
| Measured concentration (ng/mL) | RSD% | Measured concentration (ng/mL) | RSD% | Measured concentration (ng/mL) | RSD% | ||||
| ATV | 0.5 | 0.56±0.02 | 3.04 | 0.54±0.03 | 5.84 | 0.53±0.03 | 5.97 | ||
| 100 | 105.60±3.21 | 3.04 | 105.40±3.21 | 3.04 | 103.80±2.59 | 2.49 | |||
| 250 | 245.60± 9.58 | 3.90 | 247±5.52 | 2.24 | 245.80±8.81 | 3.59 | |||
| o-ATV | 0.5 | 0.46±0.01 | 1.17 | 0.46±0.01 | 1.66 | 0.46±0.004 | 0.87 | ||
| 10 | 9.34±0.35 | 3.70 | 9.64±0.21 | 2.19 | 9.64±0.19 | 1.96 | |||
| 24 | 24.17±0.86 | 3.57 | 24.18±0.81 | 3.36 | 24.24±1.02 | 4.21 | |||
| p-ATV | 0.5 | 0.53±0.02 | 4.71 | 0.50±0.003 | 0.51 | 0.53±0.02 | 3.70 | ||
| 10 | 10.43±0.28 | 2.68 | 10.32±0.19 | 1.82 | 10.43±0.13 | 1.23 | |||
| 24 | 22.86±0.61 | 2.65 | 23.12±0.80 | 3.48 | 21.73±0.22 | 0.99 | |||
表 5
Table 5 Stability of ATV, o-ATV, and p-ATV in rat plasma under various storage conditions (Mean ±SD)
| Compound | Concentration (ng/mL) | Room temperature for 2 h | 8 h at 4 ℃ | Three freezes-thraws (?80 ℃) | |||||
| Measured concentration (ng/mL) | RSD% | Measured concentration (ng/mL) | RSD% | Measured concentration (ng/mL) | RSD% | ||||
| ATV | 0.5 | 0.56±0.02 | 3.04 | 0.54±0.03 | 5.84 | 0.53±0.03 | 5.97 | ||
| 100 | 105.60±3.21 | 3.04 | 105.40±3.21 | 3.04 | 103.80±2.59 | 2.49 | |||
| 250 | 245.60± 9.58 | 3.90 | 247±5.52 | 2.24 | 245.80±8.81 | 3.59 | |||
| o-ATV | 0.5 | 0.46±0.01 | 1.17 | 0.46±0.01 | 1.66 | 0.46±0.004 | 0.87 | ||
| 10 | 9.34±0.35 | 3.70 | 9.64±0.21 | 2.19 | 9.64±0.19 | 1.96 | |||
| 24 | 24.17±0.86 | 3.57 | 24.18±0.81 | 3.36 | 24.24±1.02 | 4.21 | |||
| p-ATV | 0.5 | 0.53±0.02 | 4.71 | 0.50±0.003 | 0.51 | 0.53±0.02 | 3.70 | ||
| 10 | 10.43±0.28 | 2.68 | 10.32±0.19 | 1.82 | 10.43±0.13 | 1.23 | |||
| 24 | 22.86±0.61 | 2.65 | 23.12±0.80 | 3.48 | 21.73±0.22 | 0.99 | |||
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 127.58±43.22 | 331.06±222.726c | 108.73±23.30 | 148.92±43.53 | 224.81±123.74 | 308.87±18.87b | |
| AUC0-∞ (μg·L?1h?1) | 129.75±42.85 | 465.86±455.58c | 131.27±35.42 | 165.25±43.29 | 487.11±600.05 | 856.88±367.62b | |
| t1/2z(h) | 3.22±1.25 | 14.35±14.01c | 9.93±7.81c | 10.62±5.18 | 38.38±44.13b | 40.62±22.54b | |
| Tmax(h) | 0.23±0.18 | 0.22±0.17 | 0.13±0.05c | 0.25±0.29 | 0.62±0.82 | 0.12±0.08 | |
| Vz/F(L/kg) | 138.65±62.30 | 177.40±111.67 | 358.57±212.41c | 357.36±198.04 | 447.57±483.68 | 227.31±64.91 | |
| CLz/F (L·h?1kg?1) | 30.13±9.16 | 11.92±5.83c | 29.24±8.49 | 23.14±6.49 | 14.07±7.32 | 5.28±3.36b | |
| Cmax(μg/L) | 94.84±89.49 | 187.45±124.56c | 70.03±37.36b | 89.41±41.43 | 96.50±56.34 | 143.95±79.01 | |
表 6
Table 6 Pharmacokinetic parameters of ATV after administration (Mean ± SD, n=5)
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 127.58±43.22 | 331.06±222.726c | 108.73±23.30 | 148.92±43.53 | 224.81±123.74 | 308.87±18.87b | |
| AUC0-∞ (μg·L?1h?1) | 129.75±42.85 | 465.86±455.58c | 131.27±35.42 | 165.25±43.29 | 487.11±600.05 | 856.88±367.62b | |
| t1/2z(h) | 3.22±1.25 | 14.35±14.01c | 9.93±7.81c | 10.62±5.18 | 38.38±44.13b | 40.62±22.54b | |
| Tmax(h) | 0.23±0.18 | 0.22±0.17 | 0.13±0.05c | 0.25±0.29 | 0.62±0.82 | 0.12±0.08 | |
| Vz/F(L/kg) | 138.65±62.30 | 177.40±111.67 | 358.57±212.41c | 357.36±198.04 | 447.57±483.68 | 227.31±64.91 | |
| CLz/F (L·h?1kg?1) | 30.13±9.16 | 11.92±5.83c | 29.24±8.49 | 23.14±6.49 | 14.07±7.32 | 5.28±3.36b | |
| Cmax(μg/L) | 94.84±89.49 | 187.45±124.56c | 70.03±37.36b | 89.41±41.43 | 96.50±56.34 | 143.95±79.01 | |
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 39.86±11.09 | 133.02±18.86c | 34.16±4.44 | 42.98±10.38 | 52.86±16.54 | 33.05±13.30b | |
| AUC0-∞ (μg·L?1h?1) | 40.25±11.84 | 134.95±47.93c | 34.24±4.38 | 54.36±33.87 | 53.01±16.45 | 36.91±16.95b | |
| t1/2z(h) | 2.72±1.08 | 4.36±1.73c | 2.56±0.71 | 9.70±12.80 | 2.75±0.68 | 7.31±4.02b | |
| Tmax(h) | 2.10±2.24 | 0.28±0.14c | 1.35±0.60c | 1.85±1.39 | 1.60±0.55 | 1.95±2.35 | |
| Cmax(μg/L) | 7.53±2.00 | 52.76±14.61c | 5.90±1.81b | 7.91±3.75 | 9.95±5.92 | 6.41±5.42 | |
表 7
Table 7 Pharmacokinetic parameters of o-ATV after administration (Mean ± SD, n=5)
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 39.86±11.09 | 133.02±18.86c | 34.16±4.44 | 42.98±10.38 | 52.86±16.54 | 33.05±13.30b | |
| AUC0-∞ (μg·L?1h?1) | 40.25±11.84 | 134.95±47.93c | 34.24±4.38 | 54.36±33.87 | 53.01±16.45 | 36.91±16.95b | |
| t1/2z(h) | 2.72±1.08 | 4.36±1.73c | 2.56±0.71 | 9.70±12.80 | 2.75±0.68 | 7.31±4.02b | |
| Tmax(h) | 2.10±2.24 | 0.28±0.14c | 1.35±0.60c | 1.85±1.39 | 1.60±0.55 | 1.95±2.35 | |
| Cmax(μg/L) | 7.53±2.00 | 52.76±14.61c | 5.90±1.81b | 7.91±3.75 | 9.95±5.92 | 6.41±5.42 | |
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 26.16±17.51 | 23.18±7.42 | 10.75±1.10c | 21.00±15.08 | 17.11±3.22 | 16.24±1.77 | |
| AUC0-∞ (μg·L?1h?1) | 28.54±15.79 | 26.35±9.08 | 15.51±4.54c | 110.81±178.04 | 23.12±1.99 | 23.46±6.13 | |
| t1/2z(h) | 8.64±10.31 | 9.10±4.86 | 13.33±7.23b | 152.50±287.83 | 13.71±7.21 | 13.23±9.71 | |
| Tmax(h) | 3.10±2.70 | 0.23±0.07c | 3.60±1.67 | 2.25±1.70 | 2.80±1.10 | 4.40±1.67 | |
| Cmax(μg/L) | 12.83±15.70 | 7.89±5.70b | 0.85±0.12c | 8.10±15.28 | 1.72±0.62 | 1.51±0.67 | |
表 8
Table 8 Pharmacokinetic parameters of p-ATV after administration (Mean ± SD, n=5)
| Pharmacokinetic parameters | Normal group | High-fat diet group | |||||
| ATV | DLT+ATV | DLT (7days)+ATV | ATV | DLT+ATV | DLT (7days)+ATV | ||
| AUC0-24h (μg·L?1h?1) | 26.16±17.51 | 23.18±7.42 | 10.75±1.10c | 21.00±15.08 | 17.11±3.22 | 16.24±1.77 | |
| AUC0-∞ (μg·L?1h?1) | 28.54±15.79 | 26.35±9.08 | 15.51±4.54c | 110.81±178.04 | 23.12±1.99 | 23.46±6.13 | |
| t1/2z(h) | 8.64±10.31 | 9.10±4.86 | 13.33±7.23b | 152.50±287.83 | 13.71±7.21 | 13.23±9.71 | |
| Tmax(h) | 3.10±2.70 | 0.23±0.07c | 3.60±1.67 | 2.25±1.70 | 2.80±1.10 | 4.40±1.67 | |
| Cmax(μg/L) | 12.83±15.70 | 7.89±5.70b | 0.85±0.12c | 8.10±15.28 | 1.72±0.62 | 1.51±0.67 | |
| Group | Body weight (g) | TC (mmol/L) | TG (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) |
| High-fat diet group | 373.33±34.58 | 3.36±0.38 | 0.93±0.38 | 0.74±0.11 | 1.03±0.02 |
| ATV group | 326.75±13.84c | 2.79±0.06c | 0.72±0.27c | 1.21±0.16c | 0.83±0.04c |
| DLT group | 348.17±9.89b | 2.46±0.23c | 0.71±0.14c | 0.69±0.10 | 0.63±0.06c |
| ATV+DLT group | 261.42±21.12c | 2.34±0.19c | 0.33±0.04c | 1.45±0.10c | 0.67±0.05c |
表 9
Table 9 Serum biochemical parameters and body weight changes in different treatment groups (Mean ± SD, n=5)
| Group | Body weight (g) | TC (mmol/L) | TG (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) |
| High-fat diet group | 373.33±34.58 | 3.36±0.38 | 0.93±0.38 | 0.74±0.11 | 1.03±0.02 |
| ATV group | 326.75±13.84c | 2.79±0.06c | 0.72±0.27c | 1.21±0.16c | 0.83±0.04c |
| DLT group | 348.17±9.89b | 2.46±0.23c | 0.71±0.14c | 0.69±0.10 | 0.63±0.06c |
| ATV+DLT group | 261.42±21.12c | 2.34±0.19c | 0.33±0.04c | 1.45±0.10c | 0.67±0.05c |
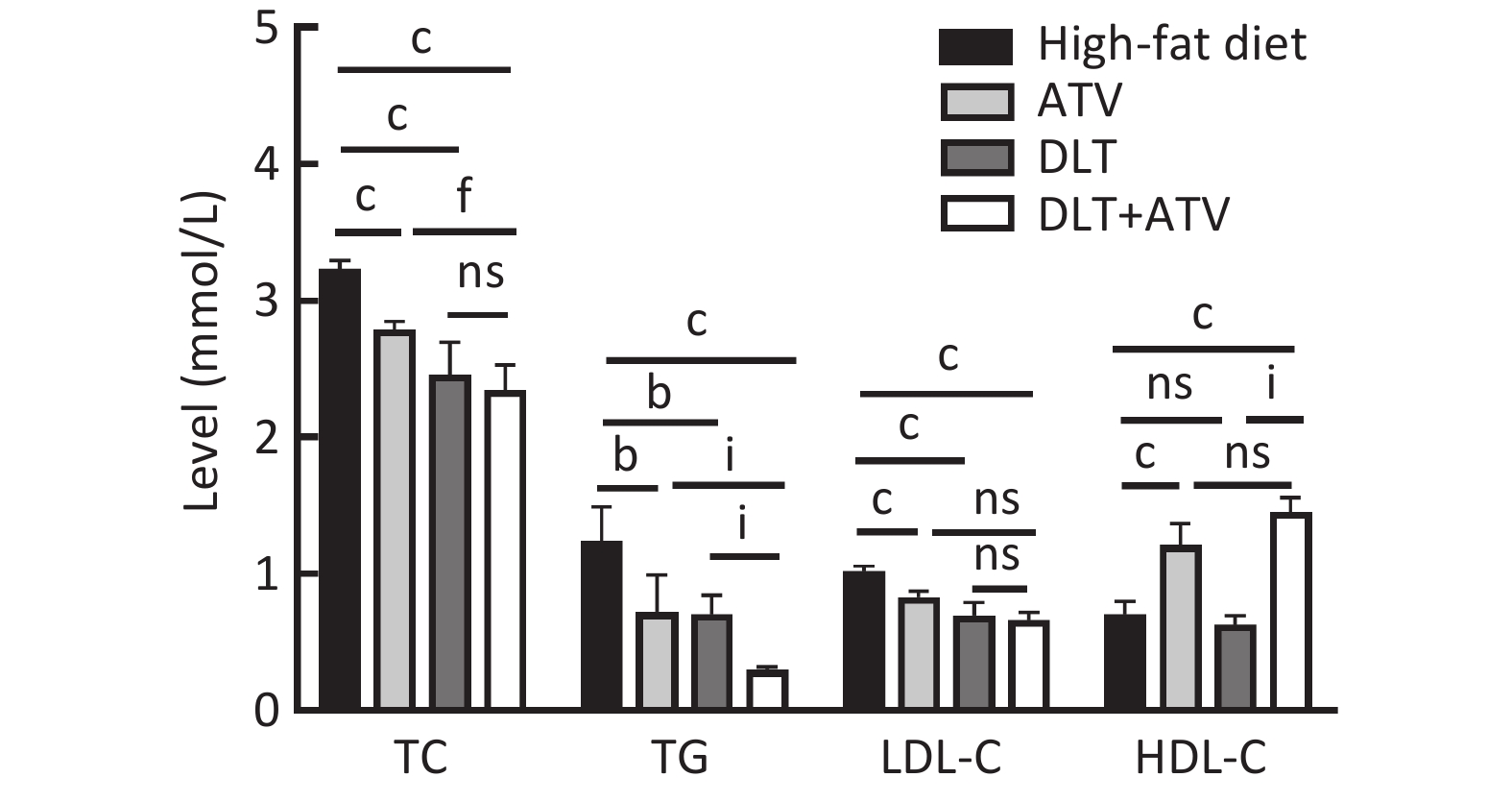
图 6
Fig.6 Serum biochemical parameters in different treatment groups (Mean ± SD, n=5) bP<0.05, cP<0.01, compared with high-fat diet group; fP<0.01, compared with atorvastatin group; iP<0.01, compared with Dan-Lou tablets group.
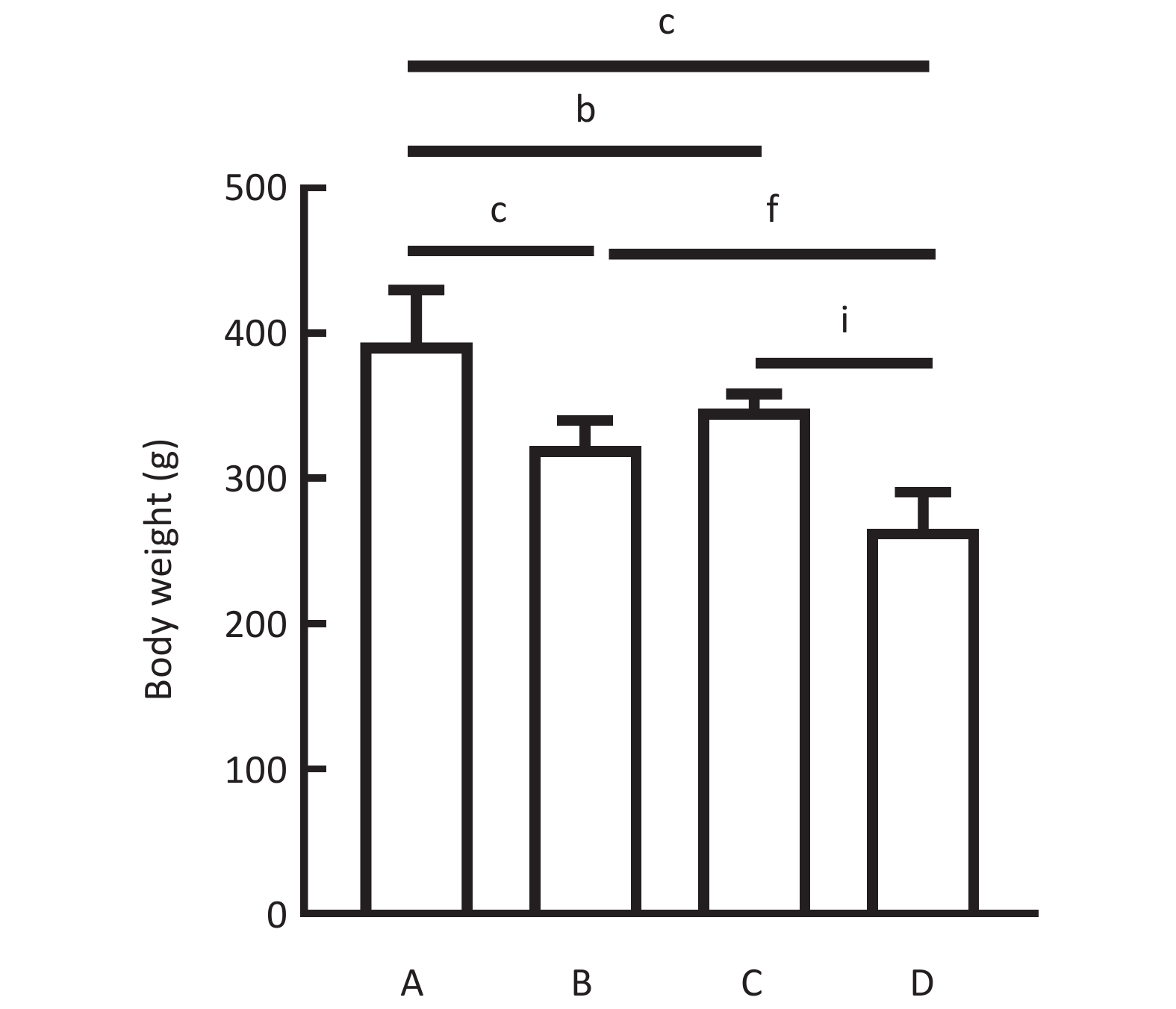
图 7
Fig.7 Body weight changes in different treatment groups (Mean ± SD, n=5) A: high fat diet group; B: atorvastatin group; C: Dan-Lou tablets group; D: atorvastatin combined with Dan-Lou tablets group. bP<0.05, cP<0.01, compared with high-fat diet group; fP<0.01, compared with atorvastatin group; iP<0.01, compared with Dan-Lou tablets group.
| 1 | Wei LL, Gu YZ, Luo Y, et al. Research progress on traditional Chinese medicine on regulating intestinal flora intreatment of cardiovascular and cerebrovascular diseases[J]. Chin Tradit Herb Drugs, 2023, 54 (18): 6185- 6194. |
| 2 | Di RN, Fan QY, Fang HL. Mechanism analysis of Chuanxiong in the treatment of cardiovascular and cerebrovascular diseases[J]. Mod Chin Med, 2022, 42, 22- 26. |
| 3 | Yu YF, Zhou ML, Luo XX, et al. Meta analysis and trial sequential analysis of colchicine in the treatment of coronary heart disease[J]. Chin Circ J, 2021, 36 (7): 659- 666. |
| 4 |
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030[J]. PLoS Med, 2006, 3 (11): e442.
doi: 10.1371/journal.pmed.0030442 |
| 5 |
Tang JJ, Li GX, Liu ZG, et al. Danlou tablet improves chronic intermittent hypoxia-induced dyslipidemia and arteriosclerosis by HIF-1alpha-Angptl4 mRNA signaling pathway[J]. Chin J Integr Med, 2022, 28 (6): 509- 517.
doi: 10.1007/s11655-020-3255-8 |
| 6 | Guo LL, Wang J, Lin F, et al. Effect of danlou tablet on arrhythmia model rats induced by transient myocardial ischemia/ reperfusion[J]. Zhongguo Zhong Xi Yi Jie He Za Zhi, 2014, 34 (9): 1125- 1129. |
| 7 |
Li Z, Cheng Q, He Y, et al. Effect of Dan-Lou tablets on coronary heart disease revealed by microarray analysis integrated with molecular mechanism studies[J]. Heliyon, 2023, 9 (5): e15777.
doi: 10.1016/j.heliyon.2023.e15777 |
| 8 |
Li Z, Yang L, Liu Y, et al. Anti-inflammatory and antioxidative effects of Dan-Lou tablets in the treatment of coronary heart disease revealed by metabolomics integrated with molecular mechanism studies[J]. J Ethnopharmacol, 2019, 240, 111911.
doi: 10.1016/j.jep.2019.111911 |
| 9 |
Mao W, Lu P, Wan R, et al. Efficacy and safety of Danlou tablets in traditional Chinese medicine for coronary heart disease: a systematic review and meta-analysis[J]. Front Cardiovasc Med, 2023, 10, 1100006.
doi: 10.3389/fcvm.2023.1100006 |
| 10 |
Ray KK, Ference BA, Séverin T, et al. World Heart Federation Cholesterol Roadmap 2022[J]. Glob Heart, 2022, 17 (1): 75.
doi: 10.5334/gh.1154 |
| 11 |
Stranberg TE, Kovanen PT, Lloyd-Jones DM, et al. Drugs for dyslipidaemia: the legacy effect of the Scandinavian Simvastatin Survival Study (4S)[J]. Lancet, 2024, 404 (10470): 2462- 2475.
doi: 10.1016/S0140-6736(24)02089-0 |
| 12 | Wang D, Li T, Tian Y, et al. Efects of atorvastatin on chronic subdural hematoma: a preliminary report from three medical centers[J]. J Neurol Sci, 2014, 336 (1/2): 237- 242. |
| 13 |
Mangione CM, Barry MJ, Nicholson WK, et al. US Preventive Services Task Force. Statin use for the primary prevention of cardiovascular disease in adults: US Preventive Services Task Force recommendation statement[J]. JAMA, 2022, 328, 746- 753.
doi: 10.1001/jama.2022.13044 |
| 14 |
Dorsch M, Kowalczyk M, Planque M, et al. Statins affect cancer cell plasticity with distinct consequences for tumor progression and metastasis[J]. Cell Rep, 2021, 37, 110056.
doi: 10.1016/j.celrep.2021.110056 |
| 15 |
Yusuf S, Bosch J, Dagenais G, et al. Cholesterol lowering in intermediate-risk persons without cardiovascular disease[J]. N Engl J Med, 2016, 374 (21): 2021- 2031.
doi: 10.1056/NEJMoa1600176 |
| 16 |
Zhang T. Physiologically based pharmacokinetic modeling of disposition and drug-drug interactions for atorvastatin and its metabolites[J]. Eur J Pharm Sci, 2015, 77, 216- 229.
doi: 10.1016/j.ejps.2015.06.019 |
| 17 |
Ding M, Ma W, Wang X, et al. A network pharmacology integrated pharmacokinetics strategy for uncovering pharmacological mechanism of compounds absorbed into the blood of Dan-Lou tablet on coronary heart disease[J]. J Ethnopharmacol, 2019, 242, 112055.
doi: 10.1016/j.jep.2019.112055 |
| 18 | Zhang Y, Guo Z, Wang J, et al. Qinlian hongqu decoction ameliorates hyperlipidemia via the IRE1-α/IKKB-β/NF-κb signaling pathway: network pharmacology and experimental validation [J]. J Ethnopharmacol, 2024, 318(Pt A): 116856. |
| 19 |
Wang L, Liu T, Zhang Q, et al. Correlation between dietary inflammation and mortality among hyperlipidemics[J]. Lipids Health Dis, 2023, 22 (1): 206.
doi: 10.1186/s12944-023-01975-0 |
| 20 |
Cholesterol Treatment Trialists' Collaboration. Effect of statin therapy on muscle symptoms: an individual participant data meta-analysis of large-scale, randomised, double-blind trials[J]. Lancet, 2022, 400 (10355): 832- 845.
doi: 10.1016/S0140-6736(22)01545-8 |
| 21 |
Izzo AA, Ernst E. Interactions between herbal medicines and prescribed drugs: an updated systematic review[J]. Drugs, 2009, 69 (13): 1777- 1798.
doi: 10.2165/11317010-000000000-00000 |
| 22 | Jiang TA. Health benefits of culinary herbs and spices[J]. J AOAC Int, 2019, 102 (2): 395- 411. |
| 23 |
安冬青, 吴宗贵, 梁春, 等. 血脂异常中西医结合诊疗专家共识[J]. 中国全科医学, 2017, 20 (3): 262- 269.
doi: 10.3969/j.issn.1007-9572.2017.03.002 |
| 24 |
Du G, Zhao H, Zhang Q, et al. A rapid method for simultaneous determination of 14 phenolic compounds in Radix puerariae using microwave-assisted extraction and ultra high performance liquid chromatography coupled with diode array detection and time-of-flight mass spectrometry[J]. J Chromatogr A, 2010, 1217, 705- 714.
doi: 10.1016/j.chroma.2009.12.017 |
| 25 |
Zhang X, Xiao HB, Xue XY, et al. Simultaneous characterization of isoflavonoids and astragalosides in two Astragalus species by high-performance liquid chromatography coupled with atmospheric pressure chemical ionization tandem mass spectrometry[J]. J Sep Sci, 2007, 30 (13): 2059- 2069.
doi: 10.1002/jssc.200700014 |
| 26 |
Yang M, Liu A, Guan S, et al. Characterization of tanshinones in the roots of Salvia miltiorrhiza (Dan-shen) by high-performance liquid chromatography with electrospray ionization tandem mass spectrometry[J]. Rapid Commun Mass Spectrom, 2006, 20 (8): 1266- 1280.
doi: 10.1002/rcm.2447 |
| 27 |
Liu AH, Guo H, Ye M, et al. Detection, characterization and identification of phenolic acids in Danshen using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry[J]. J Chromatogr, 2007, 1161 (1/2): 170- 182.
doi: 10.1016/j.chroma.2007.05.081 |
| 28 |
Liu EH, Qi LW, Peng YB, et al. Rapid separation and identification of 54 major constituents in Buyang Huanwu decoction by ultra-fast HPLC system coupled with DAD-TOF/MS[J]. Biomed Chromatogr, 2009, 23 (8): 828- 842.
doi: 10.1002/bmc.1193 |
| 29 |
Chen L, Qi J, Chang YX, et al. Identification and determination of the major constituents in Traditional Chinese Medicinal formula Danggui-Shaoyao-San by HPLC-DAD-ESI-MS/MS[J]. J Pharm Biomed Anal, 2009, 50 (2): 127- 137.
doi: 10.1016/j.jpba.2009.03.039 |
| 30 |
Liu X, Li SL, Zhou Y, et al. Characterization of protostane triterpenoids in Alisma orientalis by ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry[J]. Rapid Commun Mass Spectrom, 2010, 24 (11): 1514- 1522.
doi: 10.1002/rcm.4548 |
| 31 |
Zhao W, Huang X, Li X, et al. Qualitative and quantitative analysis of major triterpenoids in Alismatis Rhizoma by high performance liquid chromatography/diode-array detector/quadrupole-time-of-flight mass spectrometry and ultra-performance liquid chromatography/triple quadrupole mass spectrometry[J]. Molecules, 2015, 20 (8): 13958- 13981.
doi: 10.3390/molecules200813958 |
| 32 |
Chu C, Cai HX, Ren MT, et al. Characterization of novel astragaloside malonates from Radix astragali by HPLC with ESI quadrupole TOF MS[J]. J Sep Sci, 2010, 33 (4/5): 570- 581.
doi: 10.1002/jssc.200900687 |
| 33 |
Zhang XL, Liu LF, Zhu LY, et al. A high performance liquid chromatography fingerprinting and ultra high performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry chemical profiling approach to rapidly find characteristic chemical markers for quality evaluation of dispensing granules, a case study on Chuanxiong Rhizoma[J]. J Pharm Biomed Anal, 2014, 88, 391- 400.
doi: 10.1016/j.jpba.2013.09.023 |
| 34 |
Qin ZF, Dai Y, Yao Z, et al. Study on chemical profiles and metabolites of Allii Macrostemonis Bulbus as well as its representative steroidal saponins in rats by ultra-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry[J]. Food Chem, 2016, 192, 499- 515.
doi: 10.1016/j.foodchem.2015.07.040 |
| 35 |
Wang R, Zhang H, Wang Y, et al. Effects of salvianolic acid B and tanshinone IIA on the pharmacokinetics of losartan in rats by regulating the activities and expression of CYP3A4 and CYP2C9[J]. J Ethnopharmacol, 2016, 180, 87- 96.
doi: 10.1016/j.jep.2016.01.021 |
| 36 |
Paine MF, Hart HL, Ludington SS, et al. The human intestinal cytochrome P450 "pie"[J]. Drug Metab Dispos, 2006, 34 (5): 880- 886.
doi: 10.1124/dmd.105.008672 |
| 37 |
Zhang Z, Zheng H, Zhang L, et al. Atorvastatin enhances LDL receptor-mediated LDL-C uptake and modulates PCSK9 protein expression in pancreatic β-cells[J]. Islets, 2025, 17 (1): 2479906.
doi: 10.1080/19382014.2025.2479906 |
| 38 |
Zhou S, Hua S, Chen X, et al. ZeXieYin formula alleviates atherosclerosis by regulating SBAs levels through the FXR/FGF15 pathway and restoring intestinal barrier integrity[J]. Chin Med, 2025, 20 (1): 68.
doi: 10.1186/s13020-025-01116-y |
| 39 |
Lyu Q, Xue W, Liu R, et al. A brain-to-gut signal controls intestinal fat absorption[J]. Nature, 2024, 634 (8035): 936- 943.
doi: 10.1038/s41586-024-07929-5 |
| [1] | 刘媛, 崔诚, 余淼, 金文煜, 白银亮, 段雅彬, 方草, 贺建昌, 何艳, 黄桦, 霍仕霞, 金阳, 姜林, 姜哲, 焦正, 李学军, 李向阳, 李红健, 刘丽宏, 刘洋, 丘宏强, 孙凤, 孙建军, 王学昌, 王建华, 王振磊, 魏世杰, 颜晓文, 张雷, 张学农, 张宇馨, 赵军, 尹继业, 燕茹, 王新春, 刘东阳. 中国多民族人群精准用药研究价值与策略专家共识[J]. 中国临床药理学与治疗学, 2026, 31(1): 1-13. |
| [2] | 吴浩, 蒋品, 郑威, 张瑜, 左坚. LC-MS/MS法测定蛋白降解靶向嵌合体药物ARV-471在小鼠体内的浓度及其药动学[J]. 中国临床药理学与治疗学, 2025, 30(6): 774-780. |
| [3] | 宗杰, 胡瑄, 窦桂芳, 孟志云, 朱晓霞, 顾若兰, 吴卓娜, 关景丽, 甘慧. 厄他培南在老年肾功能不全患者中生理药动学模型的建立与应用[J]. 中国临床药理学与治疗学, 2025, 30(5): 622-630. |
| [4] | 孟庆恒, 韩智慧, 雷琦, 陈斌, 尹霞, 胡海棠, 刘红霞, 郑青山, 许羚, 黄钦. 基于群体药动学模型的阿立哌唑微球给药方案研究[J]. 中国临床药理学与治疗学, 2025, 30(4): 493-500. |
| [5] | 朱丽娜, 董吉, 郑利. 基于生理药代动力学模型预测肝损伤患者伏立康唑药代动力学[J]. 中国临床药理学与治疗学, 2024, 29(11): 1288-1294. |
| [6] | 查娴, 孙鲁宁, 陈潮, 王永庆. 艾司奥美拉唑在重症患者体内药代/药效学研究[J]. 中国临床药理学与治疗学, 2024, 29(10): 1152-1160. |
| [7] | 武玉洁, 赵程程, 席 庆. 蒙特卡洛模拟评价替加环素治疗革兰阴性菌感染给药方案[J]. 中国临床药理学与治疗学, 2023, 28(9): 1027-1033. |
| [8] | 李青华, 赵 艳, 赵海港, 高朋飞, 徐炳欣. ABCB1 G2677T 基因多态性检测在缺血性脑卒中患者应用阿托伐他汀降脂治疗中的价值[J]. 中国临床药理学与治疗学, 2023, 28(6): 633-640. |
| [9] | 黄志伟, 李 熠, 徐晓勇, 张 蕾, 沈一峰, 李华芳. 五种群体药动学分析工具计算结果的比较[J]. 中国临床药理学与治疗学, 2023, 28(5): 525-535. |
| [10] | 燕强勇, 向大雄, 朱荣华, 杨玲凤, 阳喜定, 李晶晶, 范晓, 刘赛, 熊守军, 方平飞. 盐酸西那卡塞片在中国健康人群的生物等效性研究[J]. 中国临床药理学与治疗学, 2023, 28(2): 171-177. |
| [11] | 刘璐, 石雨菲, 何庆烽, 徐凤艳, 王鲲, 蔡卫民, 相小强. 群体模型分析方法评估基因多态性对药物PK/PD的影响[J]. 中国临床药理学与治疗学, 2023, 28(11): 1275-1282. |
| [12] | 李雪静, 江金平, 李思凝, 万林飞, 周响响, 杨 梿, 兰 轲. 一项在中国精神分裂症受试者中评估氯氮平片的生物等效性研究[J]. 中国临床药理学与治疗学, 2023, 28(10): 1121-1130. |
| [13] | 梁美芳, 陈庆状, 杨沛群, 王勇. 基于真实世界的阿托伐他汀仿制药和原研药防治缺血性脑卒中/短暂性脑缺血发作的有效性和安全性比较[J]. 中国临床药理学与治疗学, 2022, 27(7): 785-792. |
| [14] | 张进华, 刘茂柏, 蔡铭智, 郑英丽, 劳海燕, 向倩, 都丽萍, 朱珠, 董婧, 左笑丛, 李新刚, 尚德为, 陈 冰, 叶岩荣, 王玉珠, 高建军, 张健, 陈万生, 谢海棠, 焦正. 模型引导的华法林精准用药:中国专家共识(2022版)[J]. 中国临床药理学与治疗学, 2022, 27(11): 1201-1212. |
| [15] | 徐俪颖 王淑华 张高松 黄继汉 黄小民. 和胃止泻胶囊治疗腹泻患者的临床疗效和模型化评价[J]. 中国临床药理学与治疗学, 2022, 27(1): 47-55. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||