中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (4): 543-550.doi: 10.12092/j.issn.1009-2501.2026.04.014
收稿日期:2025-05-23
修回日期:2025-09-27
出版日期:2026-04-26
发布日期:2026-04-30
通讯作者:
王月琴
E-mail:m18689718095@163.com;wangyueqin-good@163.com
作者简介:张 钰,女,硕士,研究方向:临床药理学。E-mail:基金资助:
Yu ZHANG( ), Wenbin XU, Yueqin WANG(
), Wenbin XU, Yueqin WANG( )
)
Received:2025-05-23
Revised:2025-09-27
Online:2026-04-26
Published:2026-04-30
Contact:
Yueqin WANG
E-mail:m18689718095@163.com;wangyueqin-good@163.com
摘要:
N6-甲基腺苷(N6-methyladenosine,m6A)是RNA重要的内部化学修饰,参与RNA稳定性、蛋白质翻译和前体 mRNA 剪接等RNA代谢过程,影响多种生物学过程,并在肿瘤发生发展中发挥重要作用。肿瘤免疫微环境由多种免疫细胞、细胞因子、细胞外基质等组成,影响肿瘤的生长、转移及药物治疗响应。结合m6A修饰参与多种免疫调节的作用,本文从m6A修饰直接调控免疫细胞或间接影响免疫相关因子角度,系统阐述m6A修饰与肿瘤免疫微环境之间的关联机制。
中图分类号:
张钰, 徐文斌, 王月琴. N6-甲基腺苷修饰调控肿瘤免疫微环境的研究进展[J]. 中国临床药理学与治疗学, 2026, 31(4): 543-550.
Yu ZHANG, Wenbin XU, Yueqin WANG. Progress of N6-methyladenosine modification in regulating the tumor immune microenvironment[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(4): 543-550.
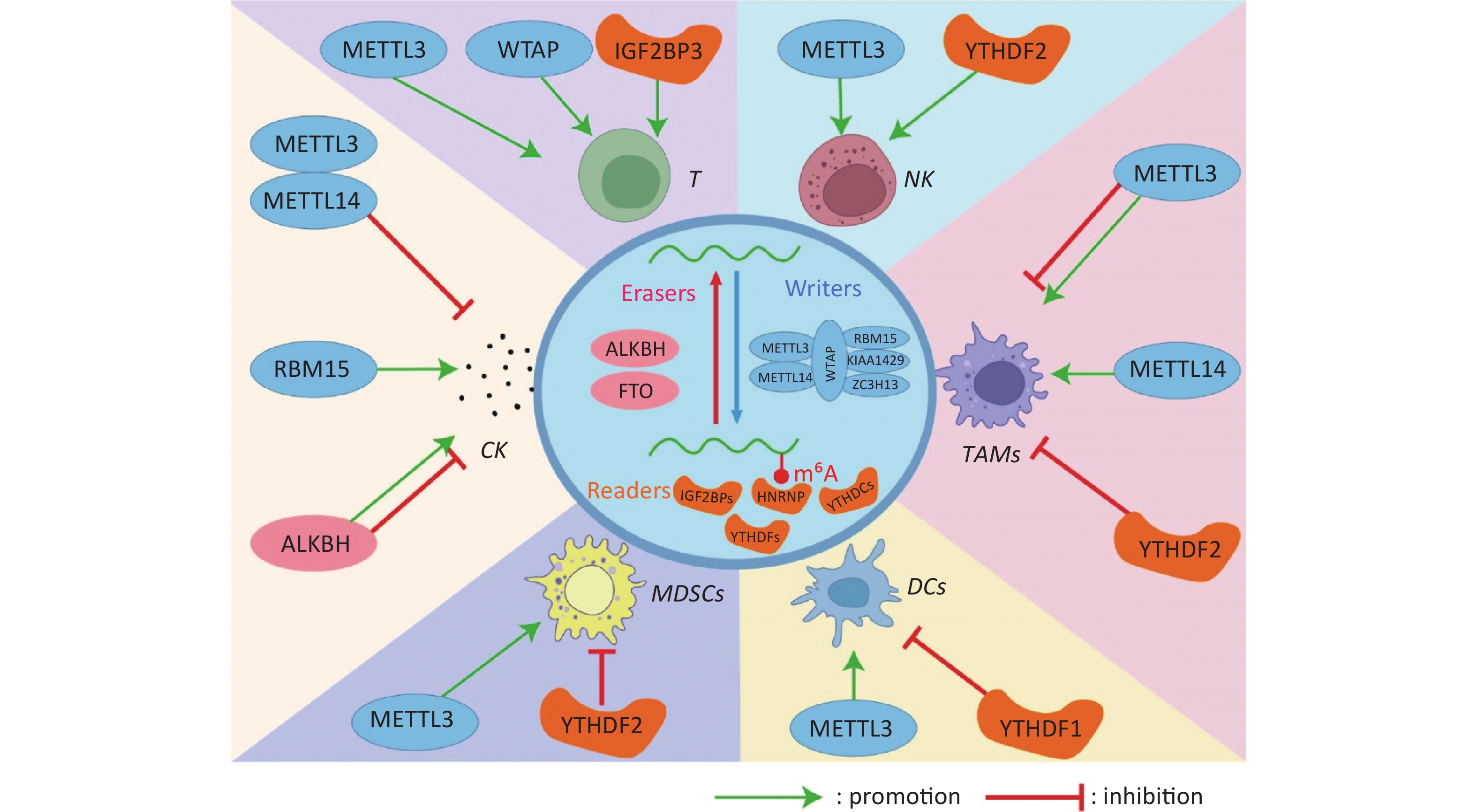
图 1 N6-甲基腺苷修饰调控肿瘤免疫微环境中免疫细胞与细胞因子
Fig.1 N6-methyladenosine modification regulates immune cells and cytokines in the tumor immune microenvironment T: T lymphocyte cell; NK: natural killer cell; DCs: dendritic cells; TAMs: tumor-associated macrophages; MDSCs: Myeloid-derived suppressor cells; CK: cytokines.
| 1 |
Bejarano L, Jordão MJC, Joyce JA. Therapeutic targeting of the tumor microenvironment[J]. Cancer Discov, 2021, 11 (4): 933- 959.
doi: 10.1158/2159-8290.CD-20-1808 |
| 2 |
Park J, Hsueh PC, Li Z, et al. Microenvironment-driven metabolic adaptations guiding CD8+ T cell anti-tumor immunity[J]. Immunity, 2023, 56 (1): 32- 42.
doi: 10.1016/j.immuni.2022.12.008 |
| 3 |
Liu Q, Gregory RI. RNAmod: an integrated system for the annotation of mRNA modifications[J]. Nucleic Acids Res, 2019, 47 (W1): W548- W555.
doi: 10.1093/nar/gkz479 |
| 4 |
吕楠, 吕志峰. m6A 修饰在慢性疼痛中的研究进展[J]. 中国疼痛医学杂志, 2023, 29 (7): 528- 534.
doi: 10.3969/j.issn.1006-9852.2023.07.008 |
| 5 | Wang X, Feng J, Xue Y, et al. Structural basis of N (6)-adenosine methylation by the METTL3-METTL14 complex[J]. Nature, 2016, 534 (7608): 575- 578. |
| 6 |
Schöller E, Weichmann F, Treiber T, et al. Interactions, localization, and phosphorylation of the m6A generating METTL3–METTL14–WTAP complex[J]. RNA, 2018, 24 (4): 499- 512.
doi: 10.1261/rna.064063.117 |
| 7 |
Coker H, Wei G, Moindrot B, et al. The role of the Xist 5' m6A region and RBM15 in X chromosome inactivation[J]. Wellcome Open Res, 2020, 5, 31.
doi: 10.12688/wellcomeopenres.15711.1 |
| 8 |
Mauer J, Sindelar M, Despic V, et al. FTO controls reversible m6Am RNA methylation during snRNA biogenesis[J]. Nat Chem Biol, 2019, 15 (4): 340- 347.
doi: 10.1038/s41589-019-0231-8 |
| 9 | 温媛媛, 黄凯玥, 史会连, 等. m6A 甲基化修饰的研究进展[J]. 生命的化学, 2022, 42 (2): 275- 282. |
| 10 | Weng H, Huang F, Yu Z, et al. The m6A reader IGF2BP2 regulates glutamine metabolism and represents a therapeutic target in acute myeloid leukemia [J]. Cancer Cell, 2022, 40 (12): 1566-1582. e10. |
| 11 |
Sun T, Wu R, Ming L. The role of m6A RNA methylation in cancer[J]. Biomed Pharmacother, 2019, 112, 108613.
doi: 10.1016/j.biopha.2019.108613 |
| 12 |
施经斌, 熊阳. 肿瘤微环境与乳腺癌细胞互作导致肿瘤转移的研究进展[J]. 中国临床药理学与治疗学, 2022, 27 (5): 562- 574.
doi: 10.12092/j.issn.1009-2501.2022.05.010 |
| 13 |
Kong F, Wang K, Wang L. Systematic analysis of the expression profile and prognostic significance of m6A regulators and PD-L1 in hepatocellular carcinoma[J]. Discover Oncol, 2022, 13 (1): 131.
doi: 10.1007/s12672-022-00595-x |
| 14 |
Xi Q, Yang G, He X, et al. M6A-mediated upregulation of lncRNA TUG1 in liver cancer cells regulates the antitumor response of CD8+ T cells and phagocytosis of macrophages[J]. Adv Sci (Weinh), 2024, 11 (34): e2400695.
doi: 10.1002/advs.202400695 |
| 15 |
Xu W, Tao M, Liu Y, et al. METTL3-mediated SMPDL3A promotes cell growth, metastasis and immune process of hepatocellular carcinoma by regulating LRPPRC[J]. Cell Signal, 2025, 127, 111543.
doi: 10.1016/j.cellsig.2024.111543 |
| 16 |
Yu F, Feng Y, Wang Q, et al. N6-methyladenosine (m6A) Writer WTAP potentiates hepatocellular carcinoma immune evasion and aerobic glycolysis[J]. Cell Biochem Biophys, 2024, 82 (3): 2321- 2331.
doi: 10.1007/s12013-024-01342-5 |
| 17 |
Chen A, Zhang VX, Zhang Q, et al. Targeting the oncogenic m6A demethylase FTO suppresses tumourigenesis and potentiates immune response in hepatocellular carcinoma[J]. Gut, 2024, 74 (1): 90- 102.
doi: 10.1136/gutjnl-2024-331903 |
| 18 |
Tsuchiya K, Yoshimura K, Inoue Y, et al. YTHDF1 and YTHDF2 are associated with better patient survival and an inflamed tumor-immune microenvironment in non-small-cell lung cancer[J]. Oncoimmunology, 2021, 10 (1): 1962656.
doi: 10.1080/2162402X.2021.1962656 |
| 19 |
Liu Z, Wang T, She Y, et al. N6-methyladenosine-modified circIGF2BP3 inhibits CD8+ T-cell responses to facilitate tumor immune evasion by promoting the deubiquitination of PD-L1 in non-small cell lung cancer[J]. Mol Cancer, 2021, 20 (1): 105.
doi: 10.1186/s12943-021-01398-4 |
| 20 |
Wang X, Chen C, Sun H, et al. m6A mRNA modification potentiates Th17 functions to inflame autoimmunity[J]. Sci China Life Sci, 2023, 66 (10): 1921- 1934.
doi: 10.1007/s11427-022-2323-4 |
| 21 |
Zhong J, Liu Z, Cai C, et al. m6A modification patterns and tumor immune landscape in clear cell renal carcinoma[J]. J Immunother Cancer, 2021, 9 (2): e001646.
doi: 10.1136/jitc-2020-001646 |
| 22 |
Tong J, Cao G, Zhang T, et al. m6A mRNA methylation sustains Treg suppressive functions[J]. Cell Res, 2018, 28 (2): 253- 256.
doi: 10.1038/cr.2018.7 |
| 23 |
Song H, Song J, Cheng M, et al. METTL3-mediated m6A RNA methylation promotes the anti-tumour immunity of natural killer cells[J]. Nat Commun, 2021, 12 (1): 5522.
doi: 10.1038/s41467-021-25803-0 |
| 24 | Wang J, Luo J, Wu X, et al. WTAP enhances the instability of SYTL1 mRNA caused by YTHDF2 in bladder cancer[J]. Histol Histopathol, 2024, 39 (5): 633- 646. |
| 25 |
Ma S, Yan J, Barr T, et al. The RNA m6A reader YTHDF2 controls NK cell antitumor and antiviral immunity[J]. J Exp Med, 2021, 218 (8): e20210279.
doi: 10.1084/jem.20210279 |
| 26 |
Wang H, Hu X, Huang M, et al. Mettl3-mediated mRNA m6A methylation promotes dendritic cell activation[J]. Nat Commun, 2019, 10 (1): 1898.
doi: 10.1038/s41467-019-09903-6 |
| 27 |
Han D, Liu J, Chen C, et al. Anti-tumour immunity controlled through mRNA m6A methylation and YTHDF1 in dendritic cells[J]. Nature, 2019, 566 (7743): 270- 274.
doi: 10.1038/s41586-019-0916-x |
| 28 |
Wen C, Wang L, Piffkó A, et al. YTHDF1 loss in dendritic cells potentiates radiation-induced antitumor immunity via STING-dependent type I IFN production[J]. J Clin Invest, 2024, 134 (23): e181612.
doi: 10.1172/JCI181612 |
| 29 |
Bai X, Wong CC, Pan Y, et al. Loss of YTHDF1 in gastric tumors restores sensitivity to antitumor immunity by recruiting mature dendritic cells[J]. J Immunother Cancer, 2022, 10 (2): e003663.
doi: 10.1136/jitc-2021-003663 |
| 30 |
Liu Y, Liu Z, Tang H, et al. The N6-methyladenosine (m6A)-forming enzyme METTL3 facilitates M1 macrophage polarization through the methylation of STAT1 mRNA[J]. Am J Physiol Cell Physiol, 2019, 317 (4): C762- C775.
doi: 10.1152/ajpcell.00212.2019 |
| 31 |
Tong J, Wang X, Liu Y, et al. Pooled CRISPR screening identifies m6A as a positive regulator of macrophage activation[J]. Sci Adv, 2021, 7 (18): eabd4742.
doi: 10.1126/sciadv.abd4742 |
| 32 |
Yin H, Zhang X, Yang P, et al. RNA m6A methylation orchestrates cancer growth and metastasis via macrophage reprogramming[J]. Nat Commun, 2021, 12 (1): 1394.
doi: 10.1038/s41467-021-21514-8 |
| 33 | Dong L, Chen C, Zhang Y, et al. The loss of RNA N6-adenosine methyltransferase Mettl14 in tumor-associated macrophages promotes CD8+ T cell dysfunction and tumor growth [J]. Cancer Cell, 2021, 39 (7): 945-957. e10. |
| 34 |
Wei H, Li W, Yang M, et al. METTL3/16-mediated m6A modification of ZNNT1 promotes hepatocellular carcinoma progression by activating ZNNT1/osteopontin/S100A9 positive feedback loop-mediated crosstalk between macrophages and tumour cells[J]. Clin Immunol, 2024, 261, 109924.
doi: 10.1016/j.clim.2024.109924 |
| 35 |
Ma S, Sun B, Duan S, et al. YTHDF2 orchestrates tumor-associated macrophage reprogramming and controls antitumor immunity through CD8+ T cells[J]. Nat Immunol, 2023, 24 (2): 255- 266.
doi: 10.1038/s41590-022-01398-6 |
| 36 |
Ye Y, Wang M, Wang G, et al. lncRNA miR4458HG modulates hepatocellular carcinoma progression by activating m6A-dependent glycolysis and promoting the polarization of tumor-associated macrophages[J]. Cell Mol Life Sci, 2023, 80 (4): 99.
doi: 10.1007/s00018-023-04741-8 |
| 37 |
Chen H, Pan Y, Zhou Q, et al. METTL3 inhibits antitumor immunity by targeting m6A-BHLHE41-CXCL1/CXCR2 axis to promote colorectal cancer[J]. Gastroenterology, 2022, 163 (4): 891- 907.
doi: 10.1053/j.gastro.2022.06.024 |
| 38 |
Ni HH, Zhang L, Huang H, et al. Connecting METTL3 and intratumoural CD33+ MDSCs in predicting clinical outcome in cervical cancer[J]. J Transl Med, 2020, 18 (1): 393.
doi: 10.1186/s12967-020-02553-z |
| 39 |
Wang J, Ling D, Shi L, et al. METTL3-mediated m6A methylation regulates ovarian cancer progression by recruiting myeloid-derived suppressor cells[J]. Cell Biosci, 2023, 13 (1): 202.
doi: 10.1186/s13578-023-01149-6 |
| 40 |
Feng L, Li M, Ma J, et al. ALKBH5 regulates arginase 1 expression in MDSCs and their immunosuppressive activity in tumor-bearing host[J]. Noncoding RNA Res, 2024, 9 (3): 913- 920.
doi: 10.1016/j.ncrna.2024.03.003 |
| 41 | Wang L, Dou X, Chen S, et al. YTHDF2 inhibition potentiates radiotherapy antitumor efficacy [J]. Cancer Cell, 2023, 41 (7): 1294-1308. e8. |
| 42 |
Wang L, Hui H, Agrawal K, et al. m6A RNA methyltransferases METTL3/14 regulate immune responses to anti-PD-1 therapy[J]. EMBO J, 2020, 39 (20): e104514.
doi: 10.15252/embj.2020104514 |
| 43 |
Mu X, Wu K, Zhu Y, et al. Intra-arterial infusion chemotherapy utilizing cisplatin inhibits bladder cancer by decreasing the fibrocytic myeloid-derived suppressor cells in an m6A-dependent manner[J]. Mol Immunol, 2021, 137, 28- 40.
doi: 10.1016/j.molimm.2021.06.012 |
| 44 |
Zeng X, Chen K, Li L, et al. Epigenetic activation of RBM15 promotes clear cell renal cell carcinoma growth, metastasis and macrophage infiltration by regulating the m6A modification of CXCL11[J]. Free Radic Biol Med, 2022, 184, 135- 147.
doi: 10.1016/j.freeradbiomed.2022.03.031 |
| 45 |
Dong F, Qin X, Wang B, et al. ALKBH5 Facilitates Hypoxia-Induced Paraspeckle Assembly and IL8 Secretion to Generate an Immunosuppressive Tumor Microenvironment[J]. Cancer Res, 2021, 81 (23): 5876- 5888.
doi: 10.1158/0008-5472.CAN-21-1456 |
| 46 |
You Y, Wen D, Zeng L, et al. ALKBH5/MAP3K8 axis regulates PD-L1+ macrophage infiltration and promotes hepatocellular carcinoma progression[J]. Int J Biol Sci, 2022, 18 (13): 5001- 5018.
doi: 10.7150/ijbs.70149 |
| 47 |
Jin S, Li M, Chang H, et al. The m6A demethylase ALKBH5 promotes tumor progression by inhibiting RIG-I expression and interferon alpha production through the IKKε/TBK1/IRF3 pathway in head and neck squamous cell carcinoma[J]. Mol Cancer, 2022, 21 (1): 97.
doi: 10.1186/s12943-022-01572-2 |
| 48 |
Wang YF, Zhang WL, Li ZX, et al. METTL14 downregulation drives S100A4+ monocyte-derived macrophages via MyD88/NF-κB pathway to promote MAFLD progression[J]. Signal Transduct Target Ther, 2024, 9 (1): 91.
doi: 10.1038/s41392-024-01797-1 |
| 49 |
Zheng H, Zheng WJ, Wang ZG, et al. Decreased expression of programmed death ligand-L1 by seven in absentia homolog 2 in cholangiocarcinoma enhances T-cell-mediated antitumor activity[J]. Front Immunol, 2022, 13, 845193.
doi: 10.3389/fimmu.2022.845193 |
| 50 | Liu Y, Liang G, Xu H, et al. Tumors exploit FTO-mediated regulation of glycolytic metabolism to evade immune surveillance [J]. Cell Metab, 2021, 33 (6): 1221-1233. e11. |
| 51 |
Wang L, Zhu L, Liang C, et al. Targeting N6-methyladenosine reader YTHDF1 with siRNA boosts antitumor immunity in NASH-HCC by inhibiting EZH2-IL-6 axis[J]. J Hepatol, 2023, 79 (5): 1185- 1200.
doi: 10.1016/j.jhep.2023.06.021 |
| 52 |
Zhang H, Luo X, Yang W, et al. YTHDF2 upregulation and subcellular localization dictate CD8 T cell polyfunctionality in anti-tumor immunity[J]. Nat Commun, 2024, 15 (1): 9559.
doi: 10.1038/s41467-024-53997-6 |
| 53 | Liu J, Zhang X, Chen K, et al. CCR7 Chemokine receptor-inducible lnc-dpf3 restrains dendritic cell migration by inhibiting HIF-1α-mediated glycolysis [J]. Immunity, 2019, 50 (3): 600-615. e15. |
| 54 |
Li Y, Xia L, Tan K, et al. N6-Methyladenosine co-transcriptionally directs the demethylation of histone H3K9me2[J]. Nat Genet, 2020, 52 (9): 870- 877.
doi: 10.1038/s41588-020-0677-3 |
| [1] | 杨永婷, 韩舒欣, 康晓静. 可切除性高危恶性黑素瘤的(新)辅助治疗研究进展[J]. 中国临床药理学与治疗学, 2025, 30(6): 849-857. |
| [2] | 杜丽娟, 林建华, 叶景焕, 宋璐, 彭燕芬, 刘玉平. 益气通腑泻热方对脂多糖诱导的小鼠急性肺损伤的作用及机制研究[J]. 中国临床药理学与治疗学, 2025, 30(5): 599-607. |
| [3] | 吴娟, 黄曦, 李佳嘉, 魏雨晴, 张丽琴, 俞咏梅, 陆志伟, 张鹤. SMARCA4缺失型非小细胞肺癌的SPP1表达及其与PD-L1的关系[J]. 中国临床药理学与治疗学, 2025, 30(4): 477-486. |
| [4] | 王龙, 王宇辰, 郭奕霖, 吴锦慧. 工程菌调控肿瘤相关巨噬细胞增强免疫治疗[J]. 中国临床药理学与治疗学, 2025, 30(3): 297-312. |
| [5] | 陈虹丹, 黄银德, 李翀. 难治性甲状腺癌药物治疗新技术和新方法的研究进展[J]. 中国临床药理学与治疗学, 2025, 30(3): 325-331. |
| [6] | 周莎莎, 程雪清, 彭冬冬, 王小青, 扶丽君, 肖文喜, 张国民. 清热消炎宁抗甲型流感H3N2病毒的药效及作用机制研究[J]. 中国临床药理学与治疗学, 2025, 30(3): 347-354. |
| [7] | 吴园园, 李晨露, 陈燕, 曹梦妲, 邵华. 替雷利珠联合仑伐替尼治疗中晚期肝癌的临床疗效分析[J]. 中国临床药理学与治疗学, 2025, 30(3): 392-397. |
| [8] | 聂扬, 汪越, 魏嘉. 靶向结合免疫激活治疗CLDN18.2阳性胃癌的研究进展[J]. 中国临床药理学与治疗学, 2025, 30(2): 146-158. |
| [9] | 陈祯美, 陈进宏. 胆管癌精准诊疗进展及前沿[J]. 中国临床药理学与治疗学, 2025, 30(2): 159-170. |
| [10] | 陈志文, 王龙蓉, 王鲁. 肝细胞癌靶向治疗的现状和进展[J]. 中国临床药理学与治疗学, 2025, 30(2): 171-182. |
| [11] | 张箫扬子, 祝永福, 梅旦. 弥漫性大B细胞淋巴瘤的病理机制及其免疫治疗的研究进展[J]. 中国临床药理学与治疗学, 2025, 30(12): 1711-1721. |
| [12] | 周智华, 常静雯, 严元元, 漆亚男, 韩晶晶, 朱欣怡, 俞晨, 吴红雁, 范方田. 参芪扶正注射液增强免疫检查点抑制剂anti-PD-L1抗肿瘤作用及机制研究[J]. 中国临床药理学与治疗学, 2024, 29(7): 792-799. |
| [13] | 白昊东, 沙炳先, Ambedkar Kumar Yadav, 徐镶怀, 余莉. 慢性阻塞性肺病的生物靶向治疗[J]. 中国临床药理学与治疗学, 2024, 29(4): 377-382. |
| [14] | 王林林, 马圆, 陈智鸿, 计海婴. 过敏原特异性免疫治疗在过敏性气道疾病中的应用进展[J]. 中国临床药理学与治疗学, 2024, 29(4): 427-431. |
| [15] | 张志鹏, 陈子琦, 田建辉. 中医药减毒增效作用在免疫检查点治疗中的应用进展[J]. 中国临床药理学与治疗学, 2024, 29(3): 339-347. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||