中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (4): 551-560.doi: 10.12092/j.issn.1009-2501.2026.04.015
收稿日期:2025-01-05
修回日期:2025-08-15
出版日期:2026-04-26
发布日期:2026-04-30
通讯作者:
曹飞
E-mail:kongzhongchenke@126.com
作者简介:张洁妤,硕士,副教授,研究方向:神经退行性疾病
基金资助:
Jieyu ZHANG, Yina WENG, Fei CAO( )
)
Received:2025-01-05
Revised:2025-08-15
Online:2026-04-26
Published:2026-04-30
Contact:
Fei CAO
E-mail:kongzhongchenke@126.com
摘要:
帕金森病(Parkinson's disease, PD)是一种年龄相关性缓慢进行性锥体外系疾病,其患病率随我国人口老龄化进程逐年增长。由于高特异性生物标志物的缺乏,PD早期诊疗仍是一个巨大挑战。微小 RNA(microRNAs,miRNAs)是一类短链非编码RNA分子,已被发现在PD患者体液中异常表达,且通过调节内源性基因参与PD病理过程。miRNAs特异性表达谱的发现使其成为具有吸引力的非侵入性生物标志物和新型治疗靶点。本文将综述miRNAs表达异常与PD诊疗关系的相关研究,并从表达谱的变化、诊断价值、治疗潜力三个方面,阐述miRNAs在PD诊疗中的研究进展。
中图分类号:
张洁妤, 翁伊娜, 曹飞. microRNAs在帕金森病患者中表达谱的变化及诊疗潜力展望[J]. 中国临床药理学与治疗学, 2026, 31(4): 551-560.
Jieyu ZHANG, Yina WENG, Fei CAO. microRNAs expression profile changes in patients with Parkinson's disease and prospects for potential diagnosis and treatment[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(4): 551-560.
| 作者 | 样本量 | 样本来源 | miRNAs | 相对 表达量 | |
| PD组 | 对照组 | ||||
| Martins et al.[ | 19 | 13 | PBMCs | miR-335, miR-374a/b, miR-199, miR-126, miR-151-5p, miR-29b/c, miR-147, miR-28-5p, miR-30b/c, miR-301a, miR-26a | 下降 |
| Zhuang et al.[ | 120 | 120 | 血浆、脑脊液 | miR-125b | 下降 |
| Pavelka et al.[ | 387 | 416 | 血浆 | miR-145-5p, miR-151a-3p, miR-145-3p, miR-130a-3p, miR- | 升高 |
| Li et al.[ | 53 | 60 | 血浆 | miR-10b-5p, miR-150-5p, miR-342-3p, miR-186-5p, miR-192-5p,miR-361-3p, miR-155-5p, miR- | 下降 |
| miR-4433b-5p, miR-335-3p, miR-130b-5p, miR-766-3p, miR-744-5p, miR- | 升高 | ||||
| Qiu et al.[ | 23 | 30 | 血浆 | miR-1976, miR-153, miR-103a, miR-29q | 升高 |
| miR-210, miR-375, miR-146a, miR-101a | 下降 | ||||
| Rai et al.[ | 16 | 16 | 血浆 | miR- miR-203b-5p, miR-205-5p, miR-708-3p, miR-106b-3p, miR-23b-3p, miR-203a-3p | 升高 |
| miR-143-5p, miR- | 下降 | ||||
| Chen et al.[ | 75 | 73 | 血浆 | miR-153, miR-223 | 下降 |
| Jang et al.[ | 5 | 20 | 血浆 | miR-195-5p, miR-495-3p, miR-23b-3p, miR-323a-3p, miR-30c-2-3p, miR-27a-3p | 升高 |
| Zhang et al.[ | 13 | 27 | 脑脊液 | miR-124 | 下降 |
| Tan et al.[ | 7 | 4 | 脑脊液 | miR-486-5p, miR-122-5p, miR-451a, miR-423-5p, let-7b-5p, miR-151a-3p, miR-320a, miR-574-5p, miR-206, miR-204-5p, miR- | 升高 |
| Tong et al.[ | 209 | 50 | 血清、脑脊液 | miR-151a-5p, miR-24, miR-485-5p, miR-331-5p, miR-214 | 升高 |
| miR-331-3p, miR-485-3p | 下降 | ||||
| Wu et al.[ | 50 | 50 | 血清 | miR-19b-3p | 下降 |
| Wan et al.[ | 51 | 51 | 血清 | miR-218-5p, miR-320-5p | 下降 |
| Aguilar et al.[ | 60 | 40 | 血清 | miR-26b-5p, miR-25-3p, miR-191-5p, miR-330, miR- | 升高 |
| Lin et al.[ | 92 | 64 | 血清 | miR*485-3p | 下降 |
| Soto et al.[ | 20 | 40 | 血清 | miR-22-3p, miR-16-5p, miR-19b-3p | 下降 |
| Manna et al.[ | 40 | 33 | 血清 | miR-22-3p, miR-223-5p | 升高 |
| Citterio et al.[ | 45 | 39 | 血清 | miR-7-1-5p, miR-223-3p | 升高 |
| Guévremont et al.[ | 287 | 168 | 血清 | miR-24-3p | 升高 |
| Cressatti et al.[ | 83 | 77 | 唾液 | miR-223, miR-153 | 下降 |
| Chen et al.[ | 30 | 330 | 唾液 | miR-874, miR-145-3p | 升高 |
表 1 帕金森病患者中微小 RNA(miRNAs)的异常表达
Table 1 Abnormal expression of miRNAs in patients with Parkinson's disease
| 作者 | 样本量 | 样本来源 | miRNAs | 相对 表达量 | |
| PD组 | 对照组 | ||||
| Martins et al.[ | 19 | 13 | PBMCs | miR-335, miR-374a/b, miR-199, miR-126, miR-151-5p, miR-29b/c, miR-147, miR-28-5p, miR-30b/c, miR-301a, miR-26a | 下降 |
| Zhuang et al.[ | 120 | 120 | 血浆、脑脊液 | miR-125b | 下降 |
| Pavelka et al.[ | 387 | 416 | 血浆 | miR-145-5p, miR-151a-3p, miR-145-3p, miR-130a-3p, miR- | 升高 |
| Li et al.[ | 53 | 60 | 血浆 | miR-10b-5p, miR-150-5p, miR-342-3p, miR-186-5p, miR-192-5p,miR-361-3p, miR-155-5p, miR- | 下降 |
| miR-4433b-5p, miR-335-3p, miR-130b-5p, miR-766-3p, miR-744-5p, miR- | 升高 | ||||
| Qiu et al.[ | 23 | 30 | 血浆 | miR-1976, miR-153, miR-103a, miR-29q | 升高 |
| miR-210, miR-375, miR-146a, miR-101a | 下降 | ||||
| Rai et al.[ | 16 | 16 | 血浆 | miR- miR-203b-5p, miR-205-5p, miR-708-3p, miR-106b-3p, miR-23b-3p, miR-203a-3p | 升高 |
| miR-143-5p, miR- | 下降 | ||||
| Chen et al.[ | 75 | 73 | 血浆 | miR-153, miR-223 | 下降 |
| Jang et al.[ | 5 | 20 | 血浆 | miR-195-5p, miR-495-3p, miR-23b-3p, miR-323a-3p, miR-30c-2-3p, miR-27a-3p | 升高 |
| Zhang et al.[ | 13 | 27 | 脑脊液 | miR-124 | 下降 |
| Tan et al.[ | 7 | 4 | 脑脊液 | miR-486-5p, miR-122-5p, miR-451a, miR-423-5p, let-7b-5p, miR-151a-3p, miR-320a, miR-574-5p, miR-206, miR-204-5p, miR- | 升高 |
| Tong et al.[ | 209 | 50 | 血清、脑脊液 | miR-151a-5p, miR-24, miR-485-5p, miR-331-5p, miR-214 | 升高 |
| miR-331-3p, miR-485-3p | 下降 | ||||
| Wu et al.[ | 50 | 50 | 血清 | miR-19b-3p | 下降 |
| Wan et al.[ | 51 | 51 | 血清 | miR-218-5p, miR-320-5p | 下降 |
| Aguilar et al.[ | 60 | 40 | 血清 | miR-26b-5p, miR-25-3p, miR-191-5p, miR-330, miR- | 升高 |
| Lin et al.[ | 92 | 64 | 血清 | miR*485-3p | 下降 |
| Soto et al.[ | 20 | 40 | 血清 | miR-22-3p, miR-16-5p, miR-19b-3p | 下降 |
| Manna et al.[ | 40 | 33 | 血清 | miR-22-3p, miR-223-5p | 升高 |
| Citterio et al.[ | 45 | 39 | 血清 | miR-7-1-5p, miR-223-3p | 升高 |
| Guévremont et al.[ | 287 | 168 | 血清 | miR-24-3p | 升高 |
| Cressatti et al.[ | 83 | 77 | 唾液 | miR-223, miR-153 | 下降 |
| Chen et al.[ | 30 | 330 | 唾液 | miR-874, miR-145-3p | 升高 |
| 作者 | miRNAs | 模型 | 治疗方法 | 功能 |
| Wang et al.[ | miR-205 | SH-SY5Y细胞 | miR-205模拟物 | 抑制 LRRK2表达 |
| Fan et al.[ | miR-153 | PD小鼠 | miR-153模拟物 | 抑制α-突触核蛋白表达 |
| He et al.[ | miR-137 | PD小鼠 | miR-137模拟物 | 抑制α-突触核蛋白表达 |
| Zhou et al.[ | miR-7 | PD小鼠 | miR-7模拟物 | 抑制NLRP3表达 |
| Li et al.[ | miR-30e | PD小鼠 | miR-30e模拟物 | 抑制NLRP3表达 |
| Zeng et al.[ | miR-135b | SH-SY5Y、PC-12细胞 | miR-135b模拟物 | 抑制NLRP3表达 |
| Hu et al.[ | miR-425 | PD小鼠 | miR-425模拟物 | 抑制RIPK1表达 |
| Wu et al.[ | miR-543-3p | PD小鼠 | miR-543-3p模拟物 | 抑制α-突触核蛋白表达 |
| Li et al.[ | miR-150 | BV2细胞 | miR-150模拟物 | 抑制促炎细胞因子表达 |
| Wang et al.[ | miR-29c-3p | PD小鼠 | miR-29c-3p模拟物 | 激活NFAT5 |
| He et al.[ | miR-100a-5p | MN9D细胞 | miR-100a-5p模拟物 | 激活Nox4/ROS/Nrf2信号通路 |
| Esfahani et al.[ | miR-101-3p | SH-SY5Y细胞 | miR-101-3p模拟物 | 促进PGC1α表达 |
| Yao et al.[ | miR-221 | PD小鼠 | miR-221模拟物 | 抑制Bim/Bax/caspase-3信号通路 |
| Esteves et al.[ | miR-124-3p | PD小鼠 | miR-124-3p模拟物 | 减少多巴胺能神经元死亡 |
| Almeida et al.[ | miR-134 | PD大鼠 | miR-134抑制剂 | 减弱纹状体多巴胺能神经元损伤 |
| Vallelunga et al.[ | miR-30c-5p | PD小鼠 | miR-30c-5p抑制剂 | 上调ATG5表达 |
| Lv et al.[ | miR-3473b | PD小鼠 | miR-3473b抑制剂 | 增加TREM2和ULK1的表达 |
| Dong et al.[ | miR-421 | PD小鼠 | miR-421抑制剂 | 抑制调节MEF2D的表达 |
| Kaurani et al.[ | miR-129-5p | PD小鼠 | miR-129-5p抑制剂 | 抑制谷氨酸转运蛋白表达 |
表 2 miRNAs与PD治疗
Table 2 miRNAs and treatment of Parkinson's disease
| 作者 | miRNAs | 模型 | 治疗方法 | 功能 |
| Wang et al.[ | miR-205 | SH-SY5Y细胞 | miR-205模拟物 | 抑制 LRRK2表达 |
| Fan et al.[ | miR-153 | PD小鼠 | miR-153模拟物 | 抑制α-突触核蛋白表达 |
| He et al.[ | miR-137 | PD小鼠 | miR-137模拟物 | 抑制α-突触核蛋白表达 |
| Zhou et al.[ | miR-7 | PD小鼠 | miR-7模拟物 | 抑制NLRP3表达 |
| Li et al.[ | miR-30e | PD小鼠 | miR-30e模拟物 | 抑制NLRP3表达 |
| Zeng et al.[ | miR-135b | SH-SY5Y、PC-12细胞 | miR-135b模拟物 | 抑制NLRP3表达 |
| Hu et al.[ | miR-425 | PD小鼠 | miR-425模拟物 | 抑制RIPK1表达 |
| Wu et al.[ | miR-543-3p | PD小鼠 | miR-543-3p模拟物 | 抑制α-突触核蛋白表达 |
| Li et al.[ | miR-150 | BV2细胞 | miR-150模拟物 | 抑制促炎细胞因子表达 |
| Wang et al.[ | miR-29c-3p | PD小鼠 | miR-29c-3p模拟物 | 激活NFAT5 |
| He et al.[ | miR-100a-5p | MN9D细胞 | miR-100a-5p模拟物 | 激活Nox4/ROS/Nrf2信号通路 |
| Esfahani et al.[ | miR-101-3p | SH-SY5Y细胞 | miR-101-3p模拟物 | 促进PGC1α表达 |
| Yao et al.[ | miR-221 | PD小鼠 | miR-221模拟物 | 抑制Bim/Bax/caspase-3信号通路 |
| Esteves et al.[ | miR-124-3p | PD小鼠 | miR-124-3p模拟物 | 减少多巴胺能神经元死亡 |
| Almeida et al.[ | miR-134 | PD大鼠 | miR-134抑制剂 | 减弱纹状体多巴胺能神经元损伤 |
| Vallelunga et al.[ | miR-30c-5p | PD小鼠 | miR-30c-5p抑制剂 | 上调ATG5表达 |
| Lv et al.[ | miR-3473b | PD小鼠 | miR-3473b抑制剂 | 增加TREM2和ULK1的表达 |
| Dong et al.[ | miR-421 | PD小鼠 | miR-421抑制剂 | 抑制调节MEF2D的表达 |
| Kaurani et al.[ | miR-129-5p | PD小鼠 | miR-129-5p抑制剂 | 抑制谷氨酸转运蛋白表达 |
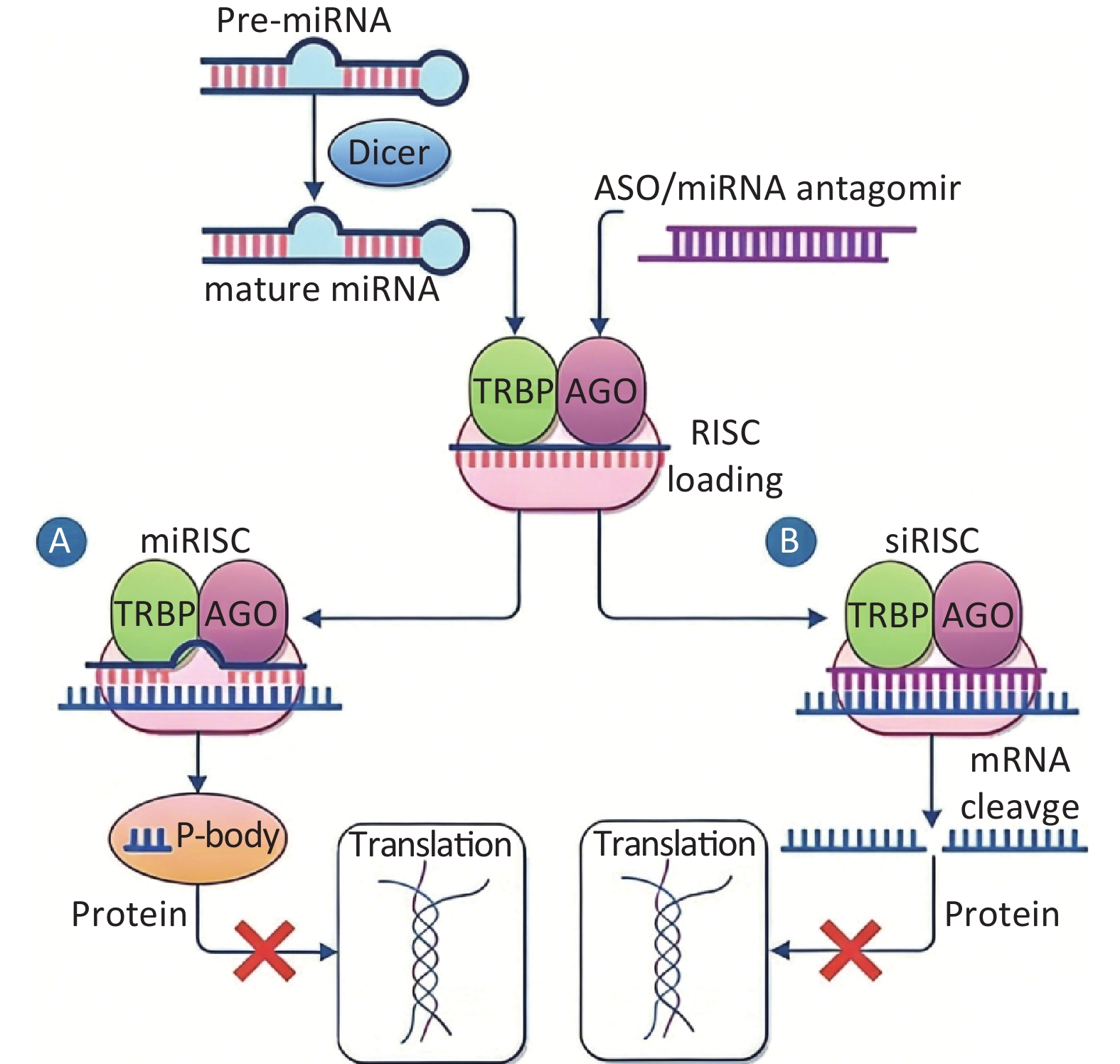
图 1 微小 RNA 抑制剂的作用机制
Fig.1 Mechanism of action of miRNA inhibitors This figure illustrates three main strategies for inhibiting miRNA function: A: anti-miRNA Oligonucleotides (AMOs) that directly bind to and degrade mature miRNAs; B: antagomirs, which are cholesterol-conjugated AMOs for enhanced in vivo stability and delivery.
| 1 |
Leitão AL, Enguita FJ. A structural view of miRNA biogenesis and function[J]. Non-coding RNA, 2022, 8 (1): 10.
doi: 10.3390/ncrna8010010 |
| 2 |
Komatsu S, Kitai H, Suzuki HI. Network regulation of microRNA biogenesis and target interaction[J]. Cells, 2023, 12 (2): 306.
doi: 10.3390/cells12020306 |
| 3 | Li S, Lei Z, Sun T. The role of microRNAs in neurodegenerative diseases: a review[J]. Cell Biol Toxicol, 2023, 39 (1): 53- 83. |
| 4 |
张舒阳, 毕研贞, 刘守胜, 等. 慢性乙型肝炎与HBV相关慢加急性肝衰竭患者血浆外泌体差异miRNA的生物信息学分析[J]. 临床肝胆病杂志, 2023, 39 (8): 1848- 1856.
doi: 10.3969/j.issn.1001-5256.2023.08.013 |
| 5 |
Martins M, Rosa A, Guedes LC, et al. Convergence of miRNA expression profiling, α-synuclein interaction and GWAS in Parkinson's disease[J]. PloS one, 2011, 6 (10): e25443.
doi: 10.1371/journal.pone.0025443 |
| 6 | Zhuang J, Cai P, Chen Z, et al. Long noncoding RNA MALAT1 and its target microRNA-125b are potential biomarkers for Alzheimer's disease management via interactions with FOXQ1, PTGS2 and CDK5[J]. Am J Transl Res, 2020, 12 (9): 5940. |
| 7 |
Pavelka L, Rauschenberger A, Hemedan A, et al. Converging peripheral blood microRNA profiles in Parkinson's disease and progressive supranuclear palsy[J]. Brain Commun, 2024, 6 (3): 187.
doi: 10.1093/braincomms/fcae187 |
| 8 |
Li Y, Cao Y, Liu W, et al. Candidate biomarkers of EV-microRNA in detecting REM sleep behavior disorder and Parkinson's disease[J]. NPJ Parkinsons Dis, 2024, 10 (1): 18.
doi: 10.1038/s41531-023-00628-4 |
| 9 |
Qiu F, Wu Y, Xie G, et al. MiRNA-1976 regulates the apoptosis of dopaminergic neurons by targeting the PINK1 gene[J]. J Integr Neurosci, 2023, 22 (2): 45.
doi: 10.31083/j.jin2202045 |
| 10 |
Rai S, Bharti PS, Singh R, et al. Circulating plasma miR-23b-3p as a biomarker target for idiopathic Parkinson's disease: comparison with small extracellular vesicle miRNA[J]. Front Neurosci, 2023, 17 (1): 1174951.
doi: 10.3389/fnins.2023.1174951 |
| 11 |
Chen W. Plasma miR-153 and miR-223 levels as potential biomarkers in Parkinson's disease[J]. Parkinsonism Relat D, 2023, 113 (1): 105512.
doi: 10.1016/j.parkreldis.2023.105596 |
| 12 |
Jang YO, Roh Y, Shin W, et al. Transferrin-conjugated magnetic nanoparticles for the isolation of brain-derived blood exosomal MicroRNAs: A novel approach for Parkinson's disease diagnosis[J]. Analytica Chimica Acta, 2024, 1306 (1): 342623.
doi: 10.1016/j.aca.2024.342623 |
| 13 |
Zhang YJ, Zhu WK, Qi FY, et al. CircHIPK3 promotes neuroinflammation through regulation of the miR-124-3p/STAT3/NLRP3 signaling pathway in Parkinson's disease[J]. Adv Clin Exp Med, 2023, 32 (3): 315- 329.
doi: 10.17219/acem/154658 |
| 14 |
Tan X, Hu J, Ming F, et al. MicroRNA-409-3p targeting at ATXN3 reduces the apoptosis of dopamine neurons based on the profile of miRNAs in the cerebrospinal fluid of early Parkinson's Disease[J]. Front Cell Dev Biol, 2022, 9, 755254.
doi: 10.3389/fcell.2021.755254 |
| 15 |
Tong G, Zhang P, Hu W, et al. Diagnostic test to Identify Parkinson's disease from the blood sera of Chinese population: A cross-sectional study[J]. Parkinsons Dis, 2022, 2022 (1): 8683877.
doi: 10.1155/2022/8683877 |
| 16 |
Wu J, Cao W, Wei W, et al. MiR-19b-3p serves as a potential diagnostic biomarker for Parkinson's disease[J]. Clin Lab, 2024, 70 (12): 2283.
doi: 10.7754/clin.lab.2024.240639 |
| 17 |
Wan Z, Rasheed M, Li Y, et al. miR-218-5p and miR-320a-5p as biomarkers for brain disorders: focus on the major depressive disorder and Parkinson's disease[J]. Mol Neurobiol, 2023, 60 (10): 5642- 5654.
doi: 10.1007/s12035-023-03391-y |
| 18 |
Aguilar MA, Ebanks S, Markus H, et al. Neuronally enriched microvesicle RNAs are differentially expressed in the serums of Parkinson's patients[J]. Front Neurosci, 2023, 17 (1): 1145923.
doi: 10.3389/fnins.2023.1145923 |
| 19 |
Lin X, Wang R, Li R, et al. Diagnostic performance of miR-485-3p in patients with Parkinson's disease and its relationship with neuroinflammation[J]. Neuromol Med, 2022, 1 (24): 195- 201.
doi: 10.1007/s12017-021-08676-w |
| 20 |
Soto M, Fernández M, Bravo P, et al. Differential serum microRNAs in premotor LRRK2 G2019S carriers from Parkinson's disease[J]. NPJ Parkinsons Dis, 2023, 9 (1): 15.
doi: 10.1038/s41531-023-00451-x |
| 21 |
Manna I, Quattrone A, Benedittis SD, et al. Exosomal miRNA as peripheral biomarkers in Parkinson's disease and progressive supranuclear palsy: A pilot study[J]. Parkinsonism & Related Disord, 2021, 93 (12): 77- 84.
doi: 10.1016/j.parkreldis.2021.11.020 |
| 22 |
Citterio LA, Mancuso R, Agostini S, et al. Serum and exosomal miR-7-1-5p and miR-223-3p as possible biomarkers for Parkinson's disease[J]. Biomolecules, 2023, 13 (5): 865.
doi: 10.3390/biom13050865 |
| 23 |
Guévremont D, Roy J, Cutfield NJ, et al. MicroRNAs in Parkinson's disease: a systematic review and diagnostic accuracy meta-analysis[J]. Scientific reports, 2023, 13 (1): 16272.
doi: 10.1038/s41598-023-43096-9 |
| 24 |
Cressatti M, Juwara L, Galindez JM, et al. Salivary microR-153 and microR‐223 levels as potential diagnostic biomarkers of idiopathic Parkinson's disease[J]. Movement Disord, 2020, 35 (3): 468- 477.
doi: 10.1002/mds.27935 |
| 25 |
Chen Y, Zheng J, Su L, et al. Increased salivary microRNAs that regulate DJ-1 gene expression as potential markers for Parkinson's disease[J]. Front Aging Neurosci, 2020, 12 (7): 210.
doi: 10.3389/fnagi.2020.00210 |
| 26 | Oliveira SR, Dionísio PA, Gaspar MM, et al. miR-335 targets LRRK2 and mitigates inflammation in Parkinson's disease[J]. Front Cell Dev Biol, 2021, 9 (4): 661461. |
| 27 |
Xu C, Bai Q, Wang C, et al. MiR-433 inhibits neuronal growth and promotes autophagy in mouse hippocampal HT-22 cell line[J]. Front Pharmacol, 2020, 11 (8): 536913.
doi: 10.3389/fphar.2020.536913 |
| 28 |
Owais S, Siddique YH. A comprehensive study of miRNAs in Parkinson's Disease: Diagnostics and therapeutic approaches[J]. CNS Neurol Disord-Dr, 2023, 22 (3): 353- 380.
doi: 10.2174/1871527321666220111152756 |
| 29 |
Thangavelu L, Moglad E, Afzal M, et al. Non-coding rnas in parkinson's disease: regulating snca and alpha-synuclein aggregation[J]. Pathol Res Pract, 2024, 9 (261): 155511.
doi: 10.1016/j.prp.2024.155511 |
| 30 | Mu C, Gao M, Xu W, et al. Mechanisms of microRNA-132 in central neurodegenerative diseases: A comprehensive review[J]. Biomed Pharmacother, 2024, 170 (1): 116029. |
| 31 |
Doroszkiewicz J, Groblewska M, Mroczko B. Molecular biomarkers and their implications for the early diagnosis of selected neurodegenerative diseases[J]. Int J Mol Sci, 2022, 23 (9): 4610.
doi: 10.3390/ijms23094610 |
| 32 | Fowler AJ, Ahn J, Hebron M, et al. CSF microRNAs reveal impairment of angiogenesis and autophagy in Parkinson disease[J]. Neurology, 2021, 7 (6): e633. |
| 33 |
Schwienbacher C, Foco L, Picard A, et al. Plasma and white blood cells show different miRNA expression profiles in Parkinson's disease[J]. J Mol Neurosci, 2017, 62 (2): 244- 254.
doi: 10.1007/s12031-017-0926-9 |
| 34 |
Soreq L, Salomonis N, Bronstein M, et al. Small RNA sequencing-microarray analyses in Parkinson leukocytes reveal deep brain stimulation-induced splicing changes that classify brain region transcriptomes[J]. Front Mol Neurosci, 2013, 6 (1): 10.
doi: 10.3389/fnmol.2013.00010 |
| 35 |
Yang Z, Li T, Cui Y, et al. Elevated plasma microRNA-105-5p level in patients with idiopathic Parkinson's disease: a potential disease biomarker[J]. Front Neurosci, 2019, 13 (3): 218.
doi: 10.3389/fnins.2019.00218 |
| 36 |
Ozdilek B, Demircan B. Serum microRNA expression levels in Turkish patients with Parkinson's disease[J]. Int J Neurosci, 2021, 131 (12): 1181- 1189.
doi: 10.1080/00207454.2020.1784165 |
| 37 |
Kim J, Inoue K, Ishii J, et al. A microRNA feedback circuit in midbrain dopamine neurons[J]. Science, 2007, 317 (5842): 1220- 1224.
doi: 10.1126/science.1140481 |
| 38 |
Minones-Moyano E, Porta S, Escaramís G, et al. MicroRNA profiling of Parkinson's disease brains identifies early downregulation of miR-34b/c which modulate mitochondrial function[J]. Hum Mol Genet, 2011, 20 (15): 3067- 3078.
doi: 10.1093/hmg/ddr210 |
| 39 |
Alvarez-Erviti L, Seow Y, Schapira AHV, et al. Influence of microRNA deregulation on chaperone-mediated autophagy and α-synuclein pathology in Parkinson's disease[J]. Cell Death Dis, 2013, 4 (3): e545- e545.
doi: 10.1038/cddis.2013.73 |
| 40 |
Cardo LF, Coto E, Ribacoba R, et al. MiRNA profile in the substantia nigra of Parkinson's disease and healthy subjects[J]. J Mol Neurosci, 2014, 54 (4): 830- 836.
doi: 10.1007/s12031-014-0428-y |
| 41 |
Bougea A. MicroRNA as candidate biomarkers in atypical Parkinsonian syndromes: systematic literature review[J]. Medicina, 2022, 58 (4): 483.
doi: 10.3390/medicina58040483 |
| 42 |
Nelson PT, Wang WX, Janse SA, et al. microRNA expression patterns in human anterior cingulate and motor cortex: A study of dementia with Lewy bodies cases and controls[J]. Brain Res, 2018, 1678 (1): 374- 383.
doi: 10.1016/j.brainres.2017.11.009 |
| 43 |
Mesarosova L, Scheper M, Iyer A, et al. miR-193b-3p/PGC-1α pathway regulates an insulin dependent anti-inflammatory response in Parkinson's disease[J]. Neurobiol Dis, 2024, 199 (1): 106587.
doi: 10.1016/j.nbd.2024.106587 |
| 44 |
Scheper M, Iyer A, Anink JJ, et al. Dysregulation of miR‐543 in Parkinson's disease: Impact on the neuroprotective gene SIRT1[J]. Neuropath Appl Neuro, 2023, 49 (1): e12864.
doi: 10.1111/nan.12864 |
| 45 |
Valente D, Zannino C, Scalise S, et al. Distinct microRNA signatures define sporadic PSP-RS and PD in patient-derived midbrain organoids[J]. iScience, 2025, 18 (1): 113162.
doi: 10.1016/j.isci.2025.113162 |
| 46 |
Yu Z, Zheng Y, Cai H, et al. Molecular beacon–based detection of circulating microRNA-containing extracellular vesicle as an α-synucleinopathy biomarker[J]. Sci Adv, 2024, 10 (20): eadl6442.
doi: 10.1126/sciadv.adl6442 |
| 47 |
Hoss AG, Labadorf A, Beach TG, et al. microRNA profiles in Parkinson's disease prefrontal cortex[J]. Front Aging Neurosci, 2016, 8 (3): 36.
doi: 10.3389/fnagi.2016.00036 |
| 48 |
Yan JH, Hua P, Chen Y, et al. Identification of microRNAs for the early diagnosis of Parkinson's disease and multiple system atrophy[J]. J Integr Neurosci, 2020, 19 (3): 429- 436.
doi: 10.31083/j.jin.2020.03.163 |
| 49 |
Soto M, Iranzo A, Lahoz S, et al. Serum MicroRNAs predict isolated rapid eye movement sleep behavior disorder and Lewy body diseases[J]. Movement Disord, 2022, 37 (10): 2086- 2098.
doi: 10.1002/mds.29171 |
| 50 |
Schulz J, Takousis P, Wohlers I, et al. Meta‐analyses identify differentially expressed microRNAs in Parkinson's disease[J]. Ann Neurol, 2019, 85 (6): 835- 851.
doi: 10.1002/ana.25490 |
| 51 |
Han L, Tang Y, Bai X, et al. Association of the serum microRNA-29 family with cognitive impairment in Parkinson's disease[J]. Aging, 2020, 12 (13): 13518.
doi: 10.18632/aging.103458 |
| 52 |
Gui YX, Liu H, Zhang LS, et al. Altered microRNA profiles in cerebrospinal fluid exosome in Parkinson disease and Alzheimer disease[J]. Oncotarget, 2015, 6 (35): 37043.
doi: 10.18632/oncotarget.6158 |
| 53 |
Botta-Orfila T, Morató X, Compta Y, et al. Identification of blood serum micro-RNAs associated with idiopathic and LRRK2 Parkinson's disease[J]. J Neurosci Res, 2014, 92 (8): 1071- 1077.
doi: 10.1002/jnr.23377 |
| 54 |
Yadav SK, Pandey A, Sarkar S, et al. Identification of altered blood MicroRNAs and plasma proteins in a rat model of Parkinson's disease[J]. Mol Neurobiol, 2022, 59 (12): 7349- 7366.
doi: 10.1007/s12035-021-02636-y |
| 55 |
Cui Y, Li T, Yang D, et al. miR-29 regulates Tet1 expression and contributes to early differentiation of mouse ESCs[J]. Oncotarget, 2016, 7 (40): 64932.
doi: 10.18632/oncotarget.10751 |
| 56 |
Maniati MS, Maniati M, Yousefi T, et al. New insights into the role of microRNAs and long noncoding RNAs in most common neurodegenerative diseases[J]. J Cell Biochem, 2019, 120 (6): 8908- 8918.
doi: 10.1002/jcb.28361 |
| 57 |
Hussain MS, Moglad E, Afzal M, et al. Autophagy‐associated non‐coding RNAs: Unraveling their impact on Parkinson's disease pathogenesis[J]. CNS Neurosci Ther, 2024, 30 (5): e14763.
doi: 10.1111/cns.14763 |
| 58 |
Wu L, Xu Q, Zhou M, et al. Plasma miR-153 and miR-223 levels as potential biomarkers in Parkinson's disease[J]. Front Neurosci, 2022, 16 (1): 865139.
doi: 10.1016/j.parkreldis.2023.105596 |
| 59 | Arora T, Sharma G, Prashar V, et al. Mechanistic evaluation of miRNAs and their targeted genes in the pathogenesis and therapeutics of Parkinson's Disease[J]. Mol Neurobiol, 2024, 61 (1): 91- 108. |
| 60 |
Paccosi E, Proietti-De-Santis L. Parkinson's disease: from genetics and epigenetics to treatment, a miRNA-based strategy[J]. Int J Mol Sci, 2023, 24 (11): 9547.
doi: 10.3390/ijms24119547 |
| 61 |
He X, Yang L, Huang R, et al. Activation of CB2R with AM1241 ameliorates neurodegeneration via the Xist/miR‐133b‐3p/Pitx3 axis[J]. J Cell Physiol, 2020, 235 (9): 6032- 6042.
doi: 10.1002/jcp.29530 |
| 62 |
Wang H, Li J, Tao L, et al. miR-205 regulates LRRK2 expression in dopamine neurons in Parkinson's disease through methylation modification[J]. Iran J Public Health, 2022, 51 (7): 1637.
doi: 10.18502/ijph.v51i7.10098 |
| 63 |
Fan TS, Liu SCH, Wu RM. Alpha-synuclein and cognitive decline in Parkinson disease[J]. Life, 2021, 11 (11): 1239.
doi: 10.3390/life11111239 |
| 64 | He M, Zhang H, Tang Z, et al. Diagnostic and therapeutic potential of exosomal microRNAs for neurodegenerative diseases[J]. Neural Plast, 2021, 2021 (1): 8884642. |
| 65 |
Zhou Y, Lu M, Du RH, et al. microRNA-7 targets nod-like receptor protein 3 inflammasome to modulate neuroinflammation in the pathogenesis of Parkinson's disease[J]. Mol Neurodegener, 2016, 11 (1): 28.
doi: 10.1186/s13024-016-0094-3 |
| 66 |
Li D, Yang H, Ma J, et al. microRNA-30e regulates neuroinflammation in MPTP model of Parkinson's disease by targeting Nlrp3[J]. Hum Cell, 2018, 31 (2): 106- 115.
doi: 10.1007/s13577-017-0187-5 |
| 67 |
Zeng R, Luo DX, Li HP, et al. microRNA-135b alleviates MPP+-mediated Parkinson's disease in in vitro model through suppressing FoxO1-induced NLRP3 inflammasome and pyroptosis[J]. J Clin Neurosci, 2019, 65 (7): 125- 133.
doi: 10.1016/j.jocn.2019.04.004 |
| 68 |
Hu YB, Zhang YF, Wang H, et al. miR-425 deficiency promotes necroptosis and dopaminergic neurodegeneration in Parkinson's disease[J]. Cell Death Dis, 2019, 10 (8): 589.
doi: 10.1038/s41419-019-1809-5 |
| 69 |
Wu X, Meng X, Tan F, et al. Regulatory mechanism of miR-543-3p on GLT-1 in a mouse model of Parkinson's disease[J]. CS Chem Neurosci, 2019, 10 (3): 1791- 1800.
doi: 10.1021/acschemneuro.8b00683 |
| 70 |
Li H, Yu L, Li M, et al. microRNA‐150 serves as a diagnostic biomarker and is involved in the inflammatory pathogenesis of Parkinson's disease[J]. Mol Genet Genom Med, 2020, 8 (4): e1189.
doi: 10.1002/mgg3.1189 |
| 71 |
Wang R, Li Q, He Y, et al. miR‐29c‐3p inhibits microglial NLRP3 inflammasome activation by targeting NFAT5 in Parkinson's disease[J]. Genes Cells, 2020, 25 (6): 364- 374.
doi: 10.1111/gtc.12764 |
| 72 |
He S, Wang Q, Chen L, et al. miR-100a-5p-enriched exosomes derived from mesenchymal stem cells enhance the anti-oxidant effect in a Parkinson's disease model via regulation of Nox4/ROS/Nrf2 signaling[J]. J Transl Med, 2023, 21 (1): 747.
doi: 10.1186/s12967-023-04638-x |
| 73 |
Esfahani MM, Mostashfi M, Hosseinabadi SV, et al. Unveiling the regulatory of miR-101-3p on ZNF746 in a Parkinson's disease cell model: Implications for therapeutic targeting[J]. Neurosci Res, 2024, 203 (1): 18- 27.
doi: 10.1016/j.neures.2023.12.001 |
| 74 |
Yao Y, Zhao Z, Zhang F, et al. microRNA-221 rescues the loss of dopaminergic neurons in a mouse model of Parkinson's disease[J]. Brain Behav, 2023, 13 (3): e2921.
doi: 10.1002/brb3.2921 |
| 75 |
Esteves M, Abreu R, Fernandes H, et al. MicroRNA-124-3p-enriched small extracellular vesicles as a therapeutic approach for Parkinson's disease[J]. Mol Ther, 2022, 30 (10): 3176- 3192.
doi: 10.1016/j.ymthe.2022.06.003 |
| 76 |
Titze de Almeida SS, Horst CH, Soto-Sánchez C, et al. Delivery of miRNA-targeted oligonucleotides in the rat striatum by magnetofection with Neuromag®[J]. Molecules, 2018, 23 (7): 1825.
doi: 10.3390/molecules23071825 |
| 77 |
Vallelunga A, Iannitti T, Dati G, et al. Serum miR-30c-5p is a potential biomarker for multiple system atrophy[J]. Mol Biol Rep, 2019, 46 (2): 1661- 1666.
doi: 10.1007/s11033-019-04614-z |
| 78 |
Lv Q, Zhong Z, Hu B, et al. microRNA‐3473b regulates the expression of TREM2/ULK1 and inhibits autophagy in inflammatory pathogenesis of Parkinson disease[J]. J Neurochem, 2021, 157 (3): 599- 610.
doi: 10.1111/jnc.15299 |
| 79 |
Dong Y, Xiong J, Ji L, et al. iR-421 aggravates neurotoxicity and promotes cell death in Parkinson's disease models by directly targeting MEF2D[J]. Neurochem Res, 2021, 46 (2): 299- 308.
doi: 10.1007/s11064-020-03166-0 |
| 80 |
Kaurani L, Zhou J, Pradhan R, et al. Inhibition of MicroRNA-129-5p promotes neuroinflammation and cognitive impairment[J]. Neurobiol Dis, 2024, 190 (1): 106361.
doi: 10.21203/rs.3.rs-3870232/v1 |
| 81 |
Stein CS, McLendon JM, Witmer NH, et al. Modulation of miR-181 influences dopaminergic neuronal degeneration in a mouse model of Parkinson's disease[J]. Mol Ther Nucl Acids, 2022, 28 (1): 1- 15.
doi: 10.1016/j.omtn.2022.02.007 |
| 82 |
Sun X, Zhang C, Tao H, et al. LINC00943 acts as miR-338-3p sponge to promote MPP+-induced SK-N-SH cell injury by directly targeting SP1 in Parkinson's disease[J]. Brain Res, 2022, 1782 (1): 147814.
doi: 10.1016/j.brainres.2022.147814 |
| 83 |
Zhou Y, Liu Y, Kang Z, et al. CircEPS15, as a sponge of MIR24-3p ameliorates neuronal damage in Parkinson disease through boosting PINK1-PRKN-mediated mitophagy[J]. Autophagy, 2023, 19 (9): 2520- 2537.
doi: 10.1080/15548627.2023.2196889 |
| [1] | 贺寿艳, 刘敏俊, 万聪, 谭布珍. PARP抑制剂治疗子宫内膜癌的研究进展[J]. 中国临床药理学与治疗学, 2026, 31(4): 536-542. |
| [2] | 张钰, 徐文斌, 王月琴. N6-甲基腺苷修饰调控肿瘤免疫微环境的研究进展[J]. 中国临床药理学与治疗学, 2026, 31(4): 543-550. |
| [3] | 张佳炜, 马雪含, 肖恩浩, 范思邈, 柳丹阳. 尿苷胞苷激酶2在肿瘤中的作用及其抑制剂研究进展[J]. 中国临床药理学与治疗学, 2026, 31(3): 362-371. |
| [4] | 张玉秀, 贺博, 张立君, 冀乐, 马壮, 郑红星, 祁珊珊. 酒精性肝病的发病机制和药物干预研究新进展[J]. 中国临床药理学与治疗学, 2026, 31(3): 409-419. |
| [5] | 陈怡伶, 朱国行. 成人癫痫药物治疗研究进展[J]. 中国临床药理学与治疗学, 2026, 31(2): 146-153. |
| [6] | 张青, 胡攀伟, 周华. 子宫粘连动物模型及其在子宫粘连研究中的应用[J]. 中国临床药理学与治疗学, 2026, 31(2): 257-264. |
| [7] | 新吉乐, 刘晶, 张欣翼, 郭家缘, 韩文卓, 孙怡馨, 赵乐, 冯卫生, 郑晓珂. 基于Connectivity Map的药物重定位评价维拉帕米作为治疗帕金森病的药物[J]. 中国临床药理学与治疗学, 2026, 31(1): 14-27. |
| [8] | 施雅妍, 王钰, 旷炜, 余守洋. 多发性硬化的发病机制和药物治疗的最新进展[J]. 中国临床药理学与治疗学, 2026, 31(1): 78-87. |
| [9] | 杜健强, 张起, 古恩鹏, 徐晨, 郭源, 张梦龙, 郭锦柯, 吴思, 谢海波. 天然化合物调控Nrf2信号通路治疗椎间盘退变的研究进展[J]. 中国临床药理学与治疗学, 2026, 31(1): 116-124. |
| [10] | 刘醒, 陈颖. 2型糖尿病的药物治疗及新技术进展[J]. 中国临床药理学与治疗学, 2025, 30(9): 1215-1223. |
| [11] | 经加吻, 孟庆波, 毕正, 王帆竞, 李瑜璠, 方朝晖. 基于治疗方法的糖尿病勃起功能障碍动物模型研究进展[J]. 中国临床药理学与治疗学, 2025, 30(9): 1224-1232. |
| [12] | 朱显军, 张丹妮, 罗喜俊, 梁俊杰, 李涛, 唐兴奎, 何嘉琳, 李伟. miR-195-5p通过靶向GPRC5A逆转胃癌细胞对曲妥珠单抗的耐药性研究[J]. 中国临床药理学与治疗学, 2025, 30(7): 929-934. |
| [13] | 杨永婷, 韩舒欣, 康晓静. 可切除性高危恶性黑素瘤的(新)辅助治疗研究进展[J]. 中国临床药理学与治疗学, 2025, 30(6): 849-857. |
| [14] | 陈冰, 左笑丛, 李新刚, 尚德为, 周佩军, 丁俊杰, 相小强, 邱晓燕, 王卓, 李晓宇, 张弋, 赵维, 王玉珠, 高建军, 焦正. 模型引导的抗排异治疗患者他克莫司精准用药专家共识[J]. 中国临床药理学与治疗学, 2025, 30(4): 433-445. |
| [15] | 吴娟, 黄曦, 李佳嘉, 魏雨晴, 张丽琴, 俞咏梅, 陆志伟, 张鹤. SMARCA4缺失型非小细胞肺癌的SPP1表达及其与PD-L1的关系[J]. 中国临床药理学与治疗学, 2025, 30(4): 477-486. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||