中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (3): 300-312.doi: 10.12092/j.issn.1009-2501.2026.03.002
王夏颖1( ), 蒋虎刚1,2, 刘佳坤1, 马静1, 刘凯1,2, 李应东1,2, 赵信科1,2,*(
), 蒋虎刚1,2, 刘佳坤1, 马静1, 刘凯1,2, 李应东1,2, 赵信科1,2,*( )
)
收稿日期:2025-05-06
修回日期:2025-06-26
出版日期:2026-03-26
发布日期:2026-04-03
通讯作者:
赵信科
E-mail:1686610858@qq.com;zxkd412@163.com
作者简介:王夏颖,女,在读硕士研究生,研究方向:中西医结合防治心血管疾病方向。E-mail:基金资助:
Xiaying WANG1( ), Hugang JIANG1,2, Jiakun LIU1, Jing MA1, Kai LIU1,2, Yingdong LI1,2, Xinke ZHAO1,2,*(
), Hugang JIANG1,2, Jiakun LIU1, Jing MA1, Kai LIU1,2, Yingdong LI1,2, Xinke ZHAO1,2,*( )
)
Received:2025-05-06
Revised:2025-06-26
Online:2026-03-26
Published:2026-04-03
Contact:
Xinke ZHAO
E-mail:1686610858@qq.com;zxkd412@163.com
摘要:
目的: 探究电离辐射对H9C2心肌细胞的影响及当归黄芪超滤物(RAS-RH)的干预效应。方法: 应用X射线建立H9C2损伤模型,并选用RAS-RH对其进行干预。通过CCK-8法检测各组H9C2的活力情况,鬼笔环肽染色观察细胞的肌丝骨架变化,JC-1染色及流式细胞术检测细胞线粒体膜电位(ΔΨm)变化,Hoechst33324染色及流式细胞术检测细胞的凋亡情况,蛋白免疫印迹法(Western blot)实验检测Drp1和HSP70相关蛋白的表达水平。结果: (1)X射线可抑制H9C2的增殖(P<0.05),破坏细胞肌丝骨架、降低细胞活力、改变ΔΨm以及促进细胞凋亡(P<0.01,P<0.05);(2)RAS-RH干预后H9C2活力升高(P<0.01)、细胞肌丝骨架恢复,且细胞的增殖能力、ΔΨm以及细胞凋亡(P<0.01、P<0.05)均得以改善;(3)RAS-RH可通过调节Drp1及HSP70蛋白的相对表达量(P<0.05)来改善H9C2生物学功能。结论: 电离辐射通过破坏细胞肌丝骨架、降低细胞活力、下调ΔΨm以及促进细胞凋亡进程来破坏H9C2的生物学功能,而RAS-RH可通过调节Drp1及HSP70蛋白的相对表达水平以修复电离辐射所带来的H9C2损伤。
中图分类号:
王夏颖, 蒋虎刚, 刘佳坤, 马静, 刘凯, 李应东, 赵信科. 当归黄芪超滤物对电离辐射诱导下心肌细胞的影响及其机制研究[J]. 中国临床药理学与治疗学, 2026, 31(3): 300-312.
Xiaying WANG, Hugang JIANG, Jiakun LIU, Jing MA, Kai LIU, Yingdong LI, Xinke ZHAO. Effect of Angelica astragalus ultrafiltration on cardiomyocytes induced by ionizing radiation and its mechanism[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(3): 300-312.
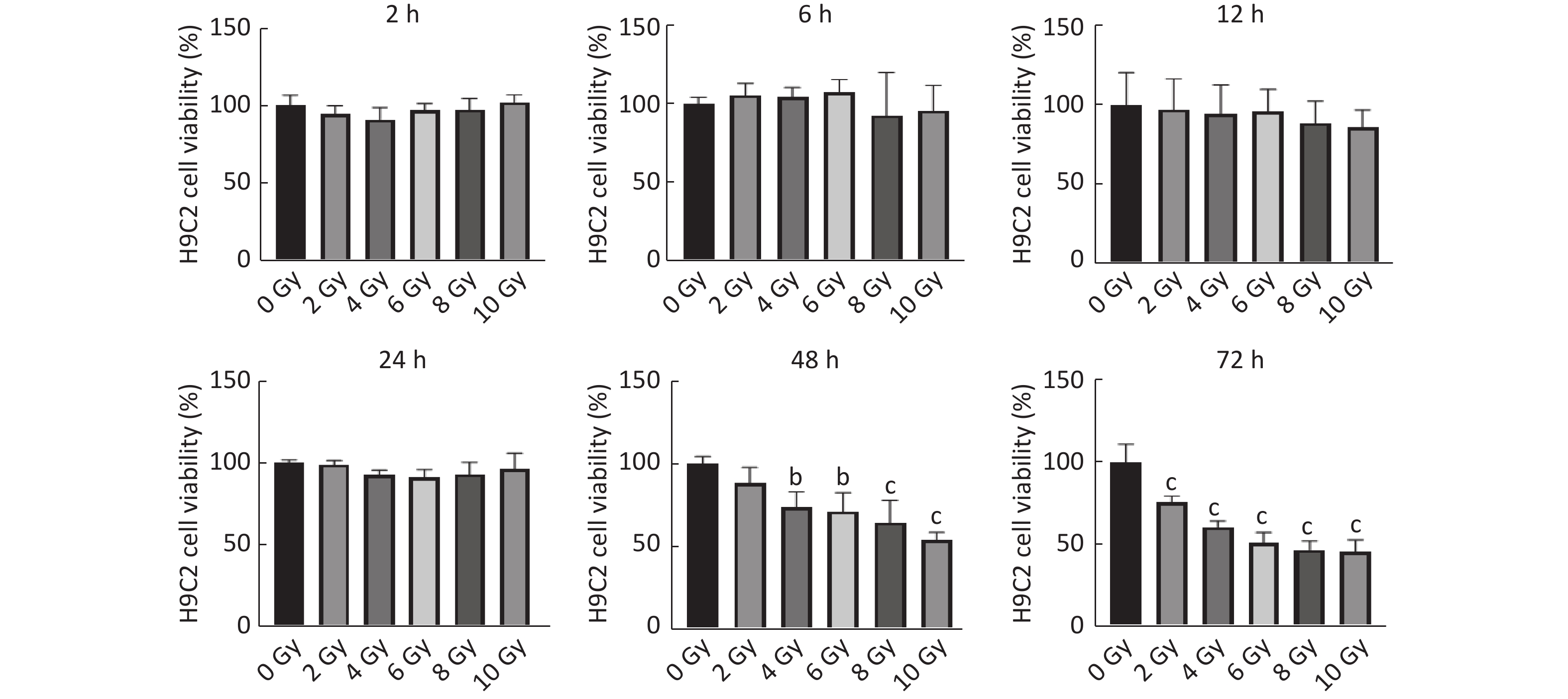
图 1
Fig.1 Effect of ionizing radiation on viability of H9C2 cells by using CCK-8 method ($ \overline{x} $±s, n=3) bP<0.05, cP<0.01, compared with 0 Gy group.

图 2
Fig.2 The effects of ionizing radiation on myofibrillar skeleton of H9C2 cells were observed by guaninyl penicillamine staining (inverted microscope, 400×)
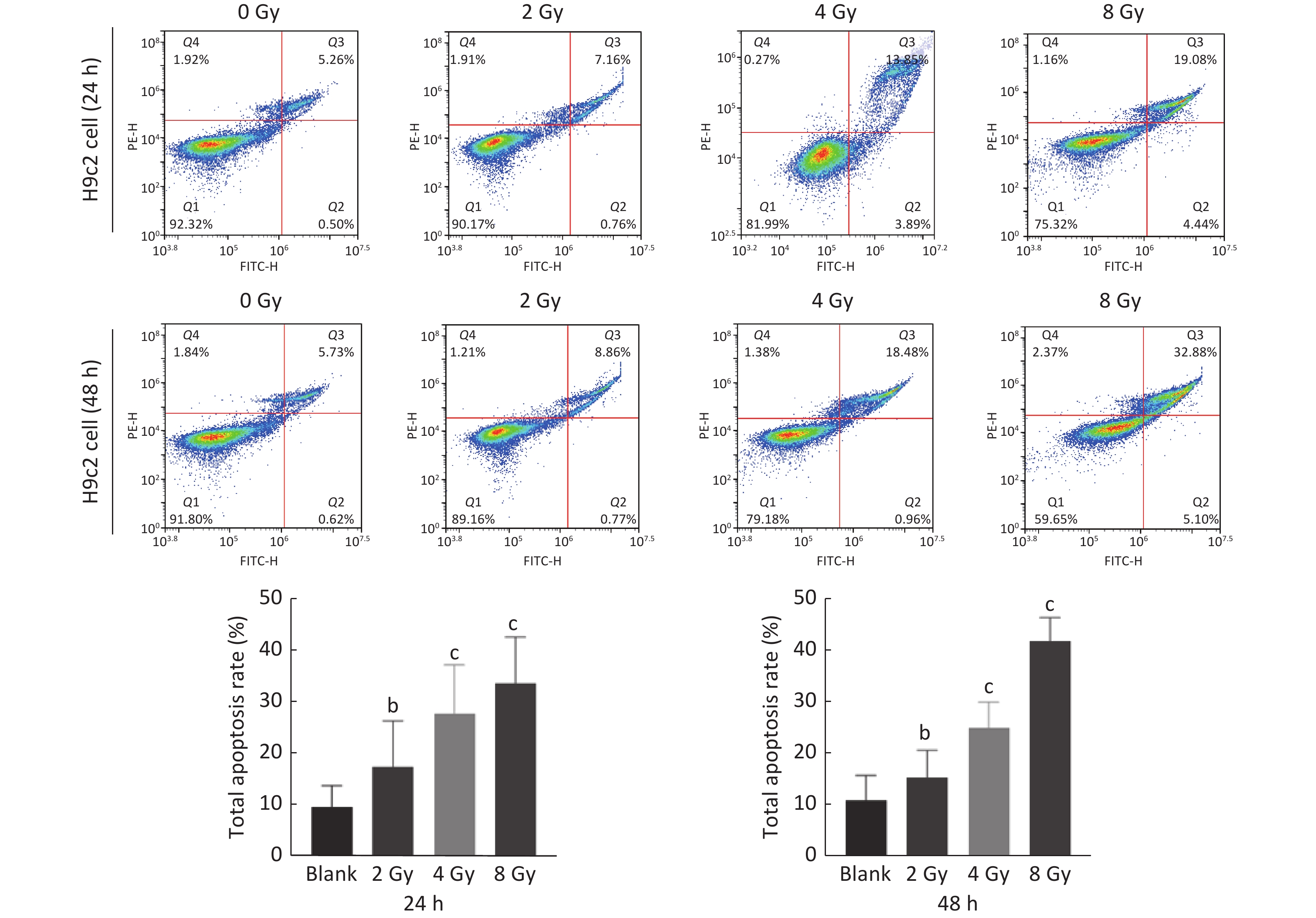
图 4
Fig.4 Effect of ionizing radiation on total apoptosis rate of H9C2 cells with the flow cytometry assay ($ \overline{x} $±s, n=3) bP<0.05, cP<0.01, compared with blank group.

图 6 cP<0.01, compared with blank group; eP<0.05, fP<0.01, compared with 2 Gy group.
Fig.6 Effect of ionizing radiation on mitochondrial transmembrane potential of H9C2 cells with the flow cytometry assay ($ \overline{x} $±s, n=3) cP<0.01, compared with blank group; eP<0.05, fP<0.01, compared with 2 Gy group.
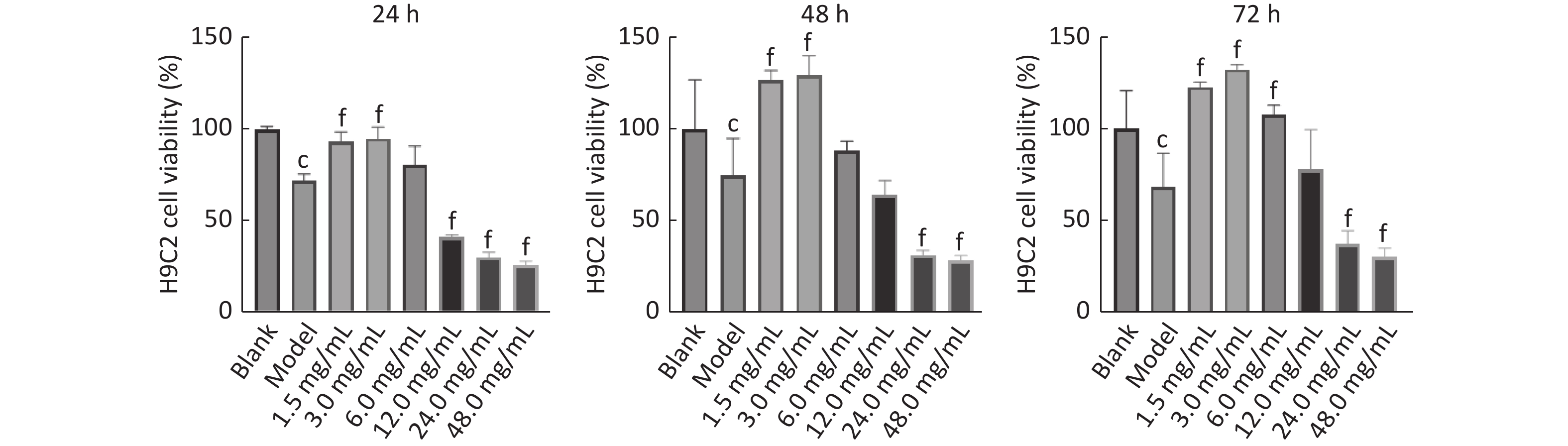
图 7
Fig.7 Effect of RAS-RH on viability of ionizing radiation-induced H9C2 cells by using CCK-8 method ($ \overline{x} $±s, n=3) cP<0.01, compared with blank group; fP<0.01, compared with model group.

图 8
Fig.8 Effect of RAS-RH on muscle silk skeleton of ionizing radiation-induced H9C2 cells by using the ghost closed-loop peptide staining (inverted microscope, 400×)
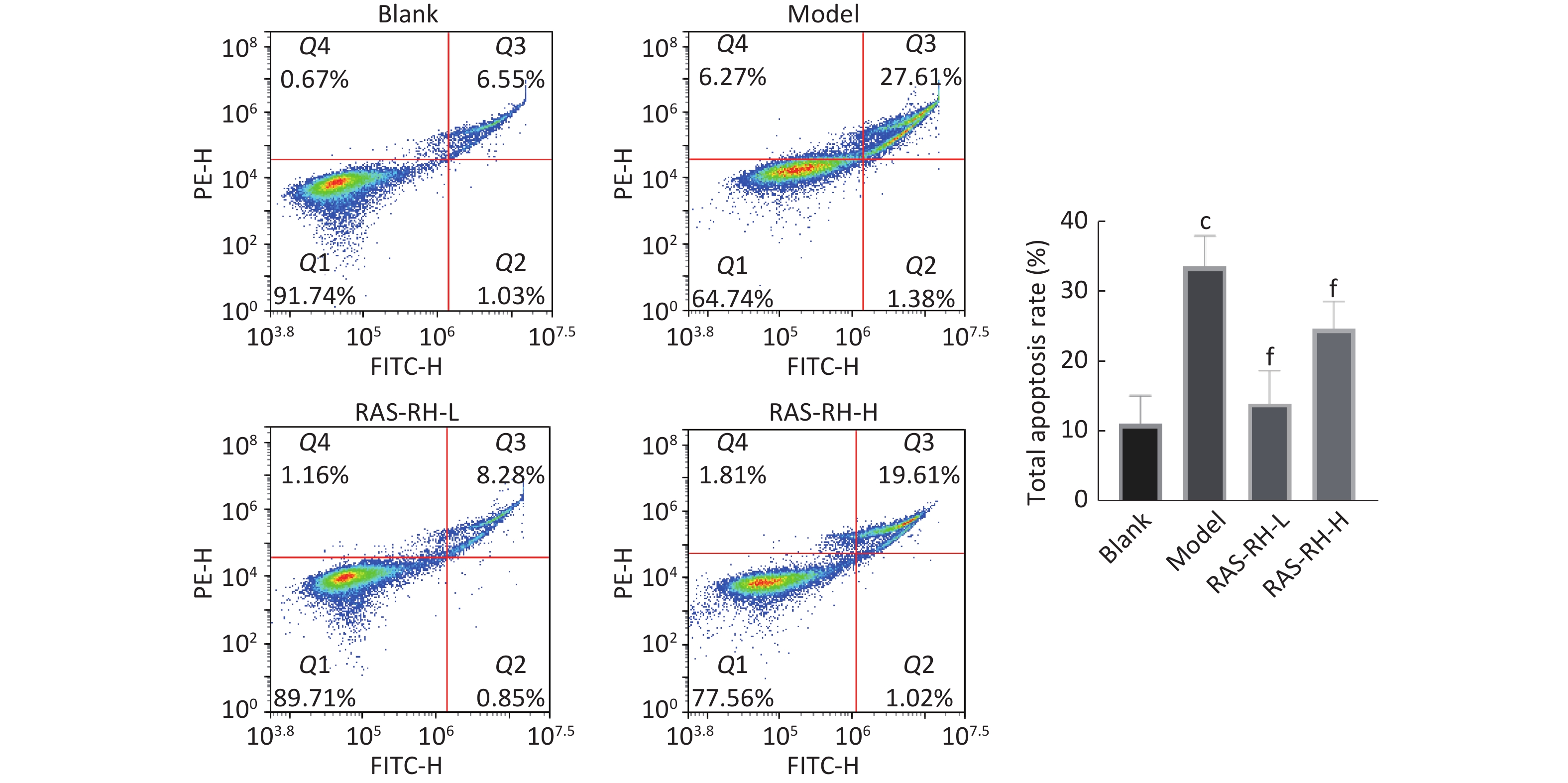
图 10
Fig.10 Effect of RAS-RH on ionizing radiation-induced apoptosis in H9C2 cells was determined by flow cytometry ($ \overline{x} $±s, n=3) cP<0.01, compared with blank group; fP<0.01, compared with model group.

图 11
Fig.11 Effect of RAS-RH on mitochondrial transmembrane potential of ionizing radiation-induced H9C2 cells by using JC-1 staining (inverted microscope, 400×)
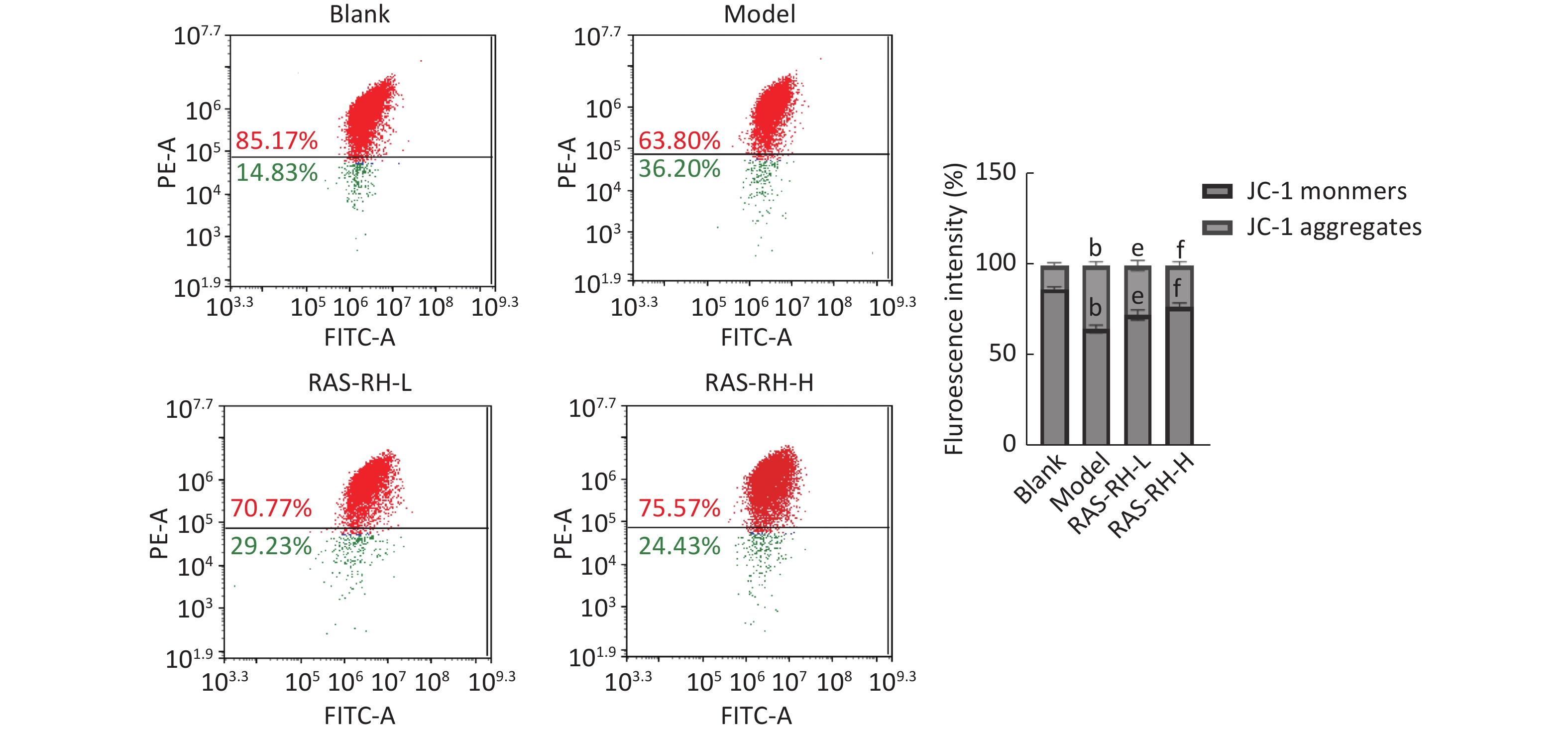
图 12 bP<0.05, compared with blank group; eP<0.05, fP<0.01, compared with model group.
Fig.12 The effect of RAS-RH on the ionizing radiation-induced mitochondrial transmembrane potential in H9C2 cells was examined by JC-1 flow cytometry ($ \overline{x} $±s, n=3) bP<0.05, compared with blank group; eP<0.05, fP<0.01, compared with model group.

图 13 bP<0.05, cP<0.01, compared with 0 Gy group.
Fig.13 Effect of Drp1 and HSP70 proteins expression in ionizing radiation-induced H9C2 cells by using Western blot technology ($ \overline{x} $±s, n=3) bP<0.05, cP<0.01, compared with 0 Gy group.
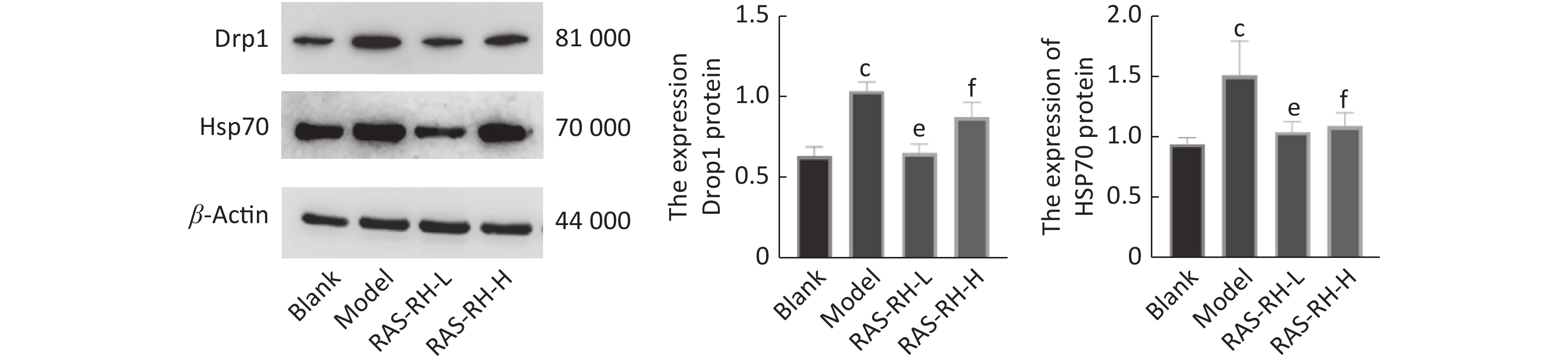
图 14 cP<0.01, compared with blank group; eP<0.05, fP<0.01, compared with model group.
Fig.14 Effect of RAS-RH on the Drp 1 and HSP70 proteins expression in ionizing radiation-induced H9C2 cells by using Western blot technology ($ \overline{x} $±s, n=3) cP<0.01, compared with blank group; eP<0.05, fP<0.01, compared with model group.
| 1 |
Viczenczova C, Kura B, Egan Benova T, et al. Irradiation-induced cardiac connexin-43 and miR-21 responses are hampered by treatment with atorvastatin and aspirin[J]. Int J Mol Sci, 2018, 19 (4): 1128.
doi: 10.3390/ijms19041128 |
| 2 |
Belzile‐Dugas E, Eisenberg MJ. Radiation‐induced cardiovascular disease: review of an underrecognized pathology[J]. J Am Heart Assoc, 2021, 10 (18): e021686.
doi: 10.1161/JAHA.121.021686 |
| 3 | Van Nimwegen FA, Schaapveld M, Janus CPM, et al. Cardiovascular disease after hodgkin lymphoma treatment: 40-year disease risk[J]. JAMA Int Med, 2015, 175 (6): 1007- 1017. |
| 4 |
Quintero-Martinez JA, Cordova-Madera SN, Villarraga HR. Radiation-induced heart disease[J]. J Clin Med, 2021, 11 (1): 146.
doi: 10.3390/jcm11010146 |
| 5 |
Sridharan V, Tripathi P, Aykin-Burns N, et al. A tocotrienol-enriched formulation protects against radiation-induced changes in cardiac mitochondria without modifying late cardiac function or structure[J]. Radiat Res, 2015, 183 (3): 357- 366.
doi: 10.1667/RR13915.1 |
| 6 |
Van Der Veen SJ, Ghobadi G, De Boer RA, et al. ACE inhibition attenuates radiation-induced cardiopulmonary damage[J]. Radiother Oncol, 2015, 114 (1): 96- 103.
doi: 10.1016/j.radonc.2014.11.017 |
| 7 |
Chistiakov DA, Shkurat TP, Melnichenko AA, et al. The role of mitochondrial dysfunction in cardiovascular disease: a brief review[J]. Ann Med, 2018, 50 (2): 121- 127.
doi: 10.1080/07853890.2017.1417631 |
| 8 |
Wang Y, Lu M, Xiong L, et al. Drp1-mediated mitochondrial fission promotes renal fibroblast activation and fibrogenesis[J]. Cell Death Dis, 2020, 11 (1): 29.
doi: 10.1038/s41419-019-2218-5 |
| 9 | Ping Z, Peng Y, Lang H, et al. Oxidative stress in radiation-induced cardiotoxicity[J]. Oxid Med Cell Longev, 2020, 2020, 3579143. |
| 10 |
Jin JY, Wei XX, Zhi XL, et al. Drp1-dependent mitochondrial fission in cardiovascular disease[J]. Acta Pharmacol Sin, 2021, 42 (5): 655- 664.
doi: 10.1038/s41401-020-00518-y |
| 11 |
Baselet B, Sonveaux P, Baatout S, et al. Pathological effects of ionizing radiation: endothelial activation and dysfunction[J]. Cell Mol Life Sci, 2019, 76 (4): 699- 728.
doi: 10.1007/s00018-018-2956-z |
| 12 |
Yin Y, Li H, Chen Y, et al. Combination of astragalus membranaceous and angelica sinensis ameliorates vascular endothelial cell dysfunction by inhibiting oxidative stress[J]. Evid Based Complement Alternat Med, 2020, 2020, 6031782.
doi: 10.1155/2020/6031782 |
| 13 | 蒋虎刚, 冯明霞, 刘凯, 等. 基于Rho/Rock通路当归红芪多糖对H9C2细胞放射性心肌损伤的影响[J]. 中国中医药信息杂志, 2019, 26 (5): 54- 58. |
| 14 |
杨飞霞, 王玉, 夏鹏飞, 等. 当归补血汤化学成分、药理作用、临床应用的研究进展及质量标志物的预测分析[J]. 中国中药杂志, 2021, 46 (11): 2677- 2685.
doi: 10.19540/j.cnki.cjcmm.20200828.201 |
| 15 |
李雯, 蒋虎刚, 王新强, 等. 当归黄芪超滤物介导Jagged1/Notch1通路抑制成纤维细胞转分化抗放射性心肌纤维化的作用机制研究[J]. 中国临床药理学与治疗学, 2025, 30 (2): 209- 215.
doi: 10.12092/j.issn.1009-2501.2025.02.007 |
| 16 |
任春贞, 袁剑芳, 王春玲, 等. 当归黄芪超滤物通过调控NLRP3/ caspase-1/ GSDMD焦亡途径改善放射性大鼠肺纤维化[J]. 中国药理学通报, 2024, 40 (11): 2124- 2131.
doi: 10.12360/CPB202403039 |
| 17 |
Chen J, Li M, Yu Y, et al. Prevention of ventricular arrhythmia complicating acute myocardial infarction by local cardiac denervation[J]. Int J Cardiol, 2015, 184, 667- 673.
doi: 10.1016/j.ijcard.2015.03.057 |
| 18 |
Darby SC, Ewert ZM, Mcgale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer[J]. N Engl J Med, 2013, 368 (11): 987- 998.
doi: 10.1056/NEJMoa1209825 |
| 19 |
Pan L, Lei D, Wang W, et al. Heart dose linked with cardiac events and overall survival in lung cancer radiotherapy: a meta-analysis[J]. Medicine (Baltimore), 2020, 99 (38): e21964.
doi: 10.1097/md.0000000000021964 |
| 20 |
Livingston K, SchlaaK RA, Puckett LL, et al. The role of mitochondrial dysfunction in radiation-induced heart disease: from bench to bedside[J]. Front Cardiovasc Med, 2020, 7, 20.
doi: 10.3389/fcvm.2020.00020 |
| 21 | 吴静, 聂祖琼, 尹琬凌. miR-499通过Drp1介导线粒体自噬保护缺氧/复氧心肌细胞[J]. 实用医学杂志, 2023, 39 (17): 2196- 2203. |
| 22 |
Liu A, Kage F, Higgs HN. Mff oligomerization is required for Drp1 activation and synergy with actin filaments during mitochondrial division[J]. Mol Biol Cell, 2021, 32 (20): ar5.
doi: 10.1091/mbc.E21-04-0224 |
| 23 | 李应东, 赵信科, 刘凯. 当归红芪超滤物对急性心肌梗死大鼠SOD、MDA、LDH1及HSP70的影响[J]. 中华中医药杂志, 2011, 26 (10): 2430- 2433. |
| 24 |
Song C, Zhang Y, Pei Q, et al. HSP70 alleviates sepsis-induced cardiomyopathy by attenuating mitochondrial dysfunction-initiated NLRP3 inflammasome- mediated pyroptosis in cardiomyocytes[J]. Burns Trauma, 2022, 10, tkac043.
doi: 10.1093/burnst/tkac043 |
| 25 |
Bhattacharya S, Asaithamby A. Ionizing radiation and heart risks[J]. Semin Cell Dev Biol, 2016, 58, 14- 25.
doi: 10.1016/j.semcdb.2016.01.045 |
| 26 |
Wang H, Wei J, Zheng Q, et al. Radiation-induced heart disease: a review of classification, mechanism and prevention[J]. Int J Biol Sci, 2019, 15 (10): 2128- 2138.
doi: 10.7150/ijbs.35460 |
| 27 |
Xie S, Sun Y, Zhao X, et al. An update of the molecular mechanisms underlying anthracycline induced cardiotoxicity[J]. Front Pharmacol, 2024, 15, 1406247.
doi: 10.3389/fphar.2024.1406247 |
| 28 |
Wu J, Chen H, Qin J, et al. Baicalin improves cardiac outcome and survival by suppressing drp1-mediated mitochondrial fission after cardiac arrest-induced myocardial damage[J]. Oxid Med Cell Longev, 2021, 2021, 8865762.
doi: 10.1155/2021/8865762 |
| 29 | Ding M, Feng N, Tang D, et al. Melatonin prevents Drp1-mediated mitochondrial fission in diabetic hearts through SIRT1-PGC1α pathway[J]. J Pineal Res, 2018, 65 (2): e12491. |
| 30 |
Havalová H, Ondrovičová G, Keresztesová B, et al. Mitochondrial HSP70 chaperone system-the influence of post-translational modifications and involvement in human diseases[J]. Int J Mol Sci, 2021, 22 (15): 8077.
doi: 10.3390/ijms22158077 |
| 31 |
Xu Z, Wang Y, Yang W, et al. Total extracts from Abelmoschus manihot (L. ) alleviate radiation-induced cardiomyocyte ferroptosis via regulating redox imbalances mediated by the NOX4/xCT/GPX4 axis[J]. J Ethnopharmacol, 2024, 334, 118582.
doi: 10.1016/j.jep.2024.118582 |
| 32 |
Farhood B, Ashrafizadeh M, Khodamoradi E, et al. Targeting of cellular redox metabolism for mitigation of radiation injury[J]. Life Sci, 2020, 250, 117570.
doi: 10.1016/j.lfs.2020.117570 |
| 33 | Wang CL, Ren CZ, Wang XY, et al. [Ultrafiltration of Angelicae Sinensis Radix and Astragali Radix inhibits ferroptosis and improves pulmonary fibrosis in rats by regulating Nrf2/xCT/GPX4 signaling pathway][J]. Zhongguo Zhong Yao Za Zhi, 2024, 49 (16): 4338- 4346. |
| 34 |
Ren C, Wang L, Li X, et al. Elucidating the mechanism of action of Radix Angelica sinensis (Oliv. ) Diels and Radix Astragalus mongholicus Bunge ultrafiltration extract on radiation-induced myocardial fibrosis based on network pharmacology and experimental research[J]. Euro J Pharmac Sci, 2024, 199, 106794.
doi: 10.1016/j.ejps.2024.106794 |
| 35 |
Ma C, Zhao X, Chang J, et al. Radix angelica sinensis and radix hedysari ultrafiltration extract protects against X-irradiation-induced cardiac fibrosis in rats[J]. Evid Based Complement Alternat Med, 2020, 2020, 4675851.
doi: 10.1155/2020/4675851 |
| [1] | 靳丽丽, 郄涛, 李立琴. 司美格鲁肽通过调控AMPK/mTOR/ULK1通路介导的自噬减轻缺氧/复氧AC16人心肌细胞炎症损伤[J]. 中国临床药理学与治疗学, 2025, 30(8): 1058-1066. |
| [2] | 李雯, 蒋虎刚, 王新强, 李应东, 刘凯, 赵信科. 当归黄芪超滤物介导Jagged1/Notch1通路抑制成纤维细胞转分化抗放射性心肌纤维化的作用机制研究[J]. 中国临床药理学与治疗学, 2025, 30(2): 209-215. |
| [3] | 郭丹丹, 杭鹏洲, 赵静, 朱华. 7,8-二羟基黄酮加剧三氧化二砷诱导的心肌细胞毒性及线粒体呼吸功能障碍[J]. 中国临床药理学与治疗学, 2025, 30(11): 1500-1507. |
| [4] | 李雯, 蒋虎刚, 王新强, 李应东, 刘凯, 赵信科. 基于网络药理学和实验验证探讨当归黄芪超滤物治疗放射性心肌纤维化的作用机制[J]. 中国临床药理学与治疗学, 2024, 29(6): 601-611. |
| [5] | 刘晓萍, 赖香茂, 欧阳资章, 江晟, 张莹. 川陈皮素抑制高糖诱导的乳鼠心肌细胞肥大[J]. 中国临床药理学与治疗学, 2021, 26(7): 753-759. |
| [6] | 徐甜甜, 吴相尧, 皮爱文, 柴惠, 张 斌, 王邦才, 窦晓兵, 朱林文思. 丹酚酸A抑制TLR4/JNK MAPK改善棕榈酸诱导的心肌细胞损伤[J]. 中国临床药理学与治疗学, 2021, 26(2): 121-128. |
| [7] | 林晓欣, 王振华. miR-29a/HMGB1信号通路在高糖高脂诱导的心肌细胞纤维化中的作用[J]. 中国临床药理学与治疗学, 2020, 25(11): 1223-1232. |
| [8] | 卓凤巧,黄占红,刘宇捷. 甘草素对高糖诱导心肌细胞凋亡的影响及机制[J]. 中国临床药理学与治疗学, 2019, 24(2): 147-151. |
| [9] | 李金玉,黄丹梅,张艳美,石刚刚,汪 彬. 巨噬细胞移动抑制因子通过调控自噬抗H9c2心肌细胞缺氧/复氧损伤[J]. 中国临床药理学与治疗学, 2018, 23(5): 531-535. |
| [10] | 郝 伟,沈媛媛,张俊秀,杨解人. 芝麻素联用维生素E对自发性高血压大鼠心肌细胞凋亡的影响[J]. 中国临床药理学与治疗学, 2018, 23(11): 1221-1227. |
| [11] | 刘 悦,刘媛媛,张 妮,曹释露,张晓京,来丽娜. Ero1α在缺氧复氧致H9C2细胞内质网应激时的表达变化及意义[J]. 中国临床药理学与治疗学, 2017, 22(9): 972-977. |
| [12] | 荆 哲, 刘峰舟, 郭文韵, 焦丕齐, 陈永清. 大蒜辣素对db/db糖尿病小鼠心肌细胞凋亡及Pink1/Parkin信号通路的影响[J]. 中国临床药理学与治疗学, 2017, 22(6): 601-605. |
| [13] | 汪和贵,张明超,胡雨龙,柯永胜. 地高辛抗血清对缺血再灌注心肌细胞凋亡的影响及机制[J]. 中国临床药理学与治疗学, 2016, 21(5): 486-489. |
| [14] | 王荣俊,丁 波,文继月,陈志武. 人urotensinⅡ对大鼠心肌细胞缺氧-再给氧损伤的保护作用及机制[J]. 中国临床药理学与治疗学, 2016, 21(12): 1341-1346. |
| [15] | 苏 丽,王 锃,黄 菲,蓝瑞隆,石 丹,陈 纯,张鲁榕,张纬建,洪金省. 甘草次酸减轻放射性炎症反应的作用机制研究[J]. 中国临床药理学与治疗学, 2016, 21(10): 1088-1094. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||