中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (2): 247-256.doi: 10.12092/j.issn.1009-2501.2026.02.012
收稿日期:2025-02-04
修回日期:2025-03-27
出版日期:2026-02-26
发布日期:2026-03-17
通讯作者:
王广基,周芳
E-mail:yuxuyou0926@163.com;gjwang@cpu.edu.cn;zf1113@163.com
作者简介:虞旭友,男,硕士,研究方向:间充质干细胞衍生细胞外囊泡用于自身免疫性疾病的研究。E-mail:基金资助:
Xuyou YU( ), Qixiang ZHANG, Jiali LIU, Guangji WANG(
), Qixiang ZHANG, Jiali LIU, Guangji WANG( ), Fang ZHOU(
), Fang ZHOU( )
)
Received:2025-02-04
Revised:2025-03-27
Online:2026-02-26
Published:2026-03-17
Contact:
Guangji WANG,Fang ZHOU
E-mail:yuxuyou0926@163.com;gjwang@cpu.edu.cn;zf1113@163.com
摘要:
炎症性肠病(inflammatory bowel disease,IBD)是一种以胃肠道慢性炎症为特征的复杂疾病,其临床治疗面临诸多挑战。尽管当前药物治疗方案在缓解临床症状方面展现出一定疗效,但长期应用仍存在感染等治疗相关风险,凸显出对新型治疗策略的迫切需求。近年来,间充质干细胞(mesenchymal stem cells,MSCs)衍生细胞外囊泡(extracellular vesicles,EVs)因其在免疫调节领域的独特优势,逐渐成为IBD治疗的研究新热点,有望成为IBD治疗领域的一种创新且有效的治疗策略。本文综述了EVs在IBD治疗领域中的最新进展,主要包括EVs的质量控制标准、免疫调节机制以及工程化策略等内容。本综述旨在为MSC-EVs在IBD的临床转化治疗领域提供参考。同时,探讨了通过工程化手段增强MSC-EVs药效的新策略,为该领域的研究开辟了创新思路。
中图分类号:
虞旭友, 张启祥, 刘嘉莉, 王广基, 周芳. 间充质干细胞衍生细胞外囊泡用于炎症性肠病的研究进展[J]. 中国临床药理学与治疗学, 2026, 31(2): 247-256.
Xuyou YU, Qixiang ZHANG, Jiali LIU, Guangji WANG, Fang ZHOU. Research progress on the application of extracellular vesicles derived from mesenchymal stem cells for inflammatory bowel disease[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(2): 247-256.
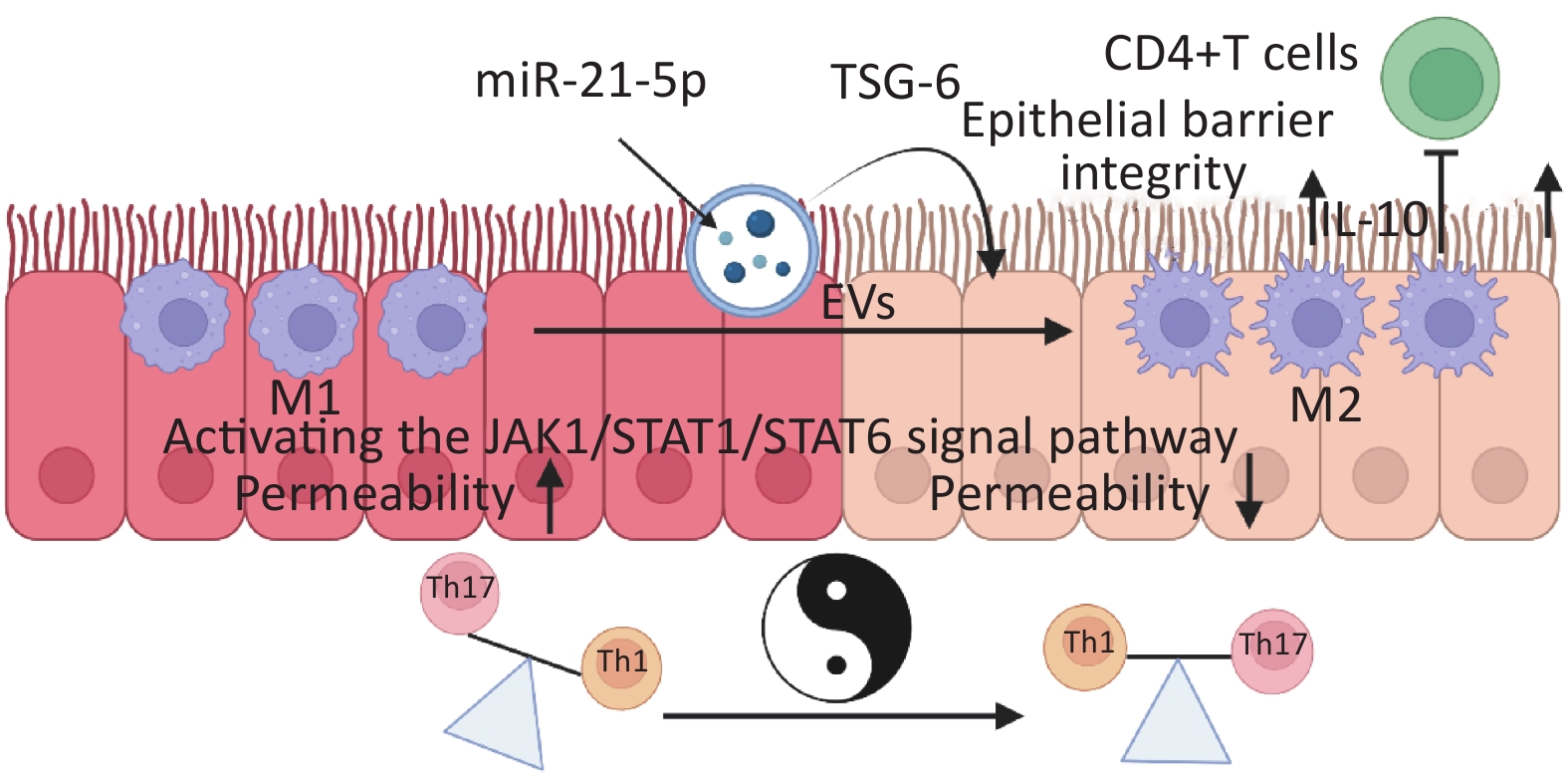
图 2 MSC-EVs治疗IBD的机制图,图片由BioRender创作
Fig.2 Mechanism for the treatment of IBD with MSC-EVs. The figure was created with BioRender (https://BioRender.com)
| 作用机制 | 动物模型 | 给药剂量及频次 | 给药途径 | 参考文献 |
| 通过JAK1/STAT1/ STAT6通路促进M2巨噬细胞复极化 | DSS小鼠 | 7天给药50 μg MSC-EVs | i.p. | [ |
| 通过miR-378a-5p/NLRP3轴调控巨噬细胞焦亡 | DSS小鼠 | 3、6、9天给药1 mg MSC-EVs | i.v. | [ |
| 通过SPRY2/ERK轴促进M2巨噬细胞复极化 | DSS小鼠 | 给药200 μg MSC-EVs | 肠系膜注射 (mesenteric injection) | [ |
| 保护肠道屏障的完整性,激活肠上皮细胞和肠道干细胞增殖 | DSS小鼠 | 0、3、6天给药300 μg MSC-EVs | i.v. | [ |
| 激活Wnt信号通路,显著促进ISCs的增殖与分化,从而加速肠道上皮的再生 | DSS及TNBS小鼠 | 4、6、8天给药300 μg MSC-EVs | i.p. | [ |
| 通过TSG-6修复小鼠黏膜屏障并维持肠道稳态 | DSS及TNBS小鼠 | 第5天给药200 μg MSC-EVs | i.p. | [ |
| 促使M2b巨噬细胞极化,诱导产生IL-10抑制CD4+T细胞 | DSS小鼠 | 2、4、6天给药100 μg MSC-EVs | i.p. | [ |
| 调整Th1/Th17的比例并促进诱导Treg细胞的表达 | DSS小鼠 | 2、4天给药60 μg MSC-EVs | i.v. | [ |
表 1 MSC-EVs在炎症性肠病动物模型中的研究
Table 1 MSC-EVs in IBD animal models
| 作用机制 | 动物模型 | 给药剂量及频次 | 给药途径 | 参考文献 |
| 通过JAK1/STAT1/ STAT6通路促进M2巨噬细胞复极化 | DSS小鼠 | 7天给药50 μg MSC-EVs | i.p. | [ |
| 通过miR-378a-5p/NLRP3轴调控巨噬细胞焦亡 | DSS小鼠 | 3、6、9天给药1 mg MSC-EVs | i.v. | [ |
| 通过SPRY2/ERK轴促进M2巨噬细胞复极化 | DSS小鼠 | 给药200 μg MSC-EVs | 肠系膜注射 (mesenteric injection) | [ |
| 保护肠道屏障的完整性,激活肠上皮细胞和肠道干细胞增殖 | DSS小鼠 | 0、3、6天给药300 μg MSC-EVs | i.v. | [ |
| 激活Wnt信号通路,显著促进ISCs的增殖与分化,从而加速肠道上皮的再生 | DSS及TNBS小鼠 | 4、6、8天给药300 μg MSC-EVs | i.p. | [ |
| 通过TSG-6修复小鼠黏膜屏障并维持肠道稳态 | DSS及TNBS小鼠 | 第5天给药200 μg MSC-EVs | i.p. | [ |
| 促使M2b巨噬细胞极化,诱导产生IL-10抑制CD4+T细胞 | DSS小鼠 | 2、4、6天给药100 μg MSC-EVs | i.p. | [ |
| 调整Th1/Th17的比例并促进诱导Treg细胞的表达 | DSS小鼠 | 2、4天给药60 μg MSC-EVs | i.v. | [ |
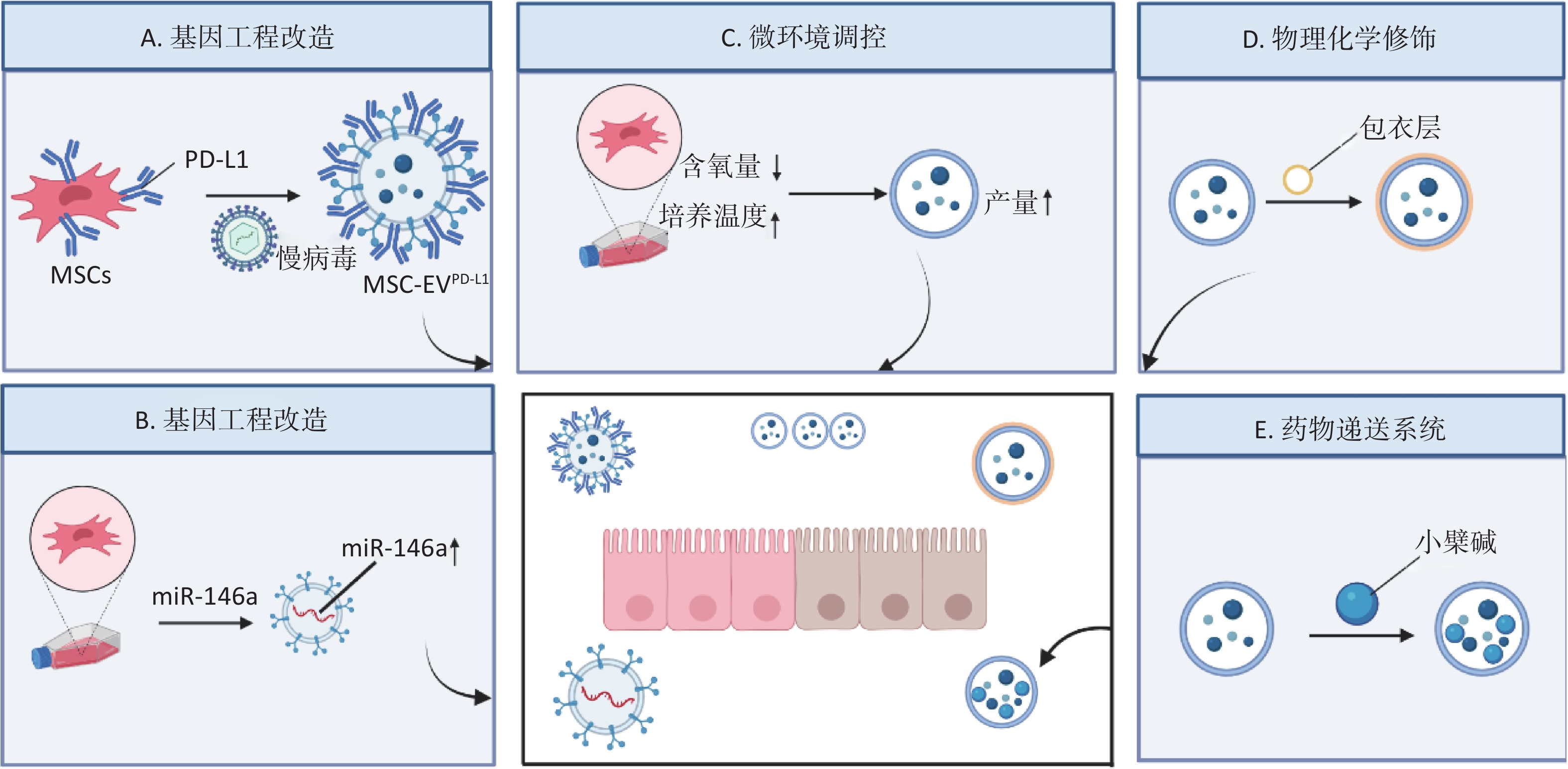
图 3 工程化MSC-EVs治疗IBD的多元化策略,图片由BioRender创作 A:高表达PD-L1的MSCs来源的EVs;B:高表达miR-146a的MSCs来源的EVs;C:微环境调控促进MSC-EVs分泌;D:经HTCC-OKGM包衣的MSC-EVs特定靶向于结肠;E:MSC-EVs负载Ber构建药物递送系统。
Fig.3 Diverse Strategies for the Treatment of IBD with engineered MSC-EVs. The figure was created with BioRender (https://BioRender.com)
| 策略 | EVs来源 | 特点 | 药效作用 | 参考文献 |
| 免疫调节 | 小鼠骨髓MSCs来源EVs | 高表达PD-L1的MSC-EVs | 重塑免疫微环境 | [ |
| 人牙周韧带MSCs来源EVs | 热应激提升MSC-EVs产量 | 增强免疫调节作用 | [ | |
| 增加药效与 递送效率 | 人脐带MSCs来源EVs | 包封MSC-EVs的微胶囊系统 | 提升MSC-EVs的稳定性、靶向性及药效 | [ |
| 人胎盘MSCs来源EVs | 结肠特异性递送 | 靶向结肠并可控释放MSC-EVs | [ | |
| 人胎盘MSCs来源EVs | MSC-EVs负载Ber | 提高药物溶解度和生物利用度 | [ | |
| 人骨髓MSCs来源EVs | MSC-EVs负载Wnt激动剂 | 增强药物积累和生物利用度 | [ | |
| 人牙周韧带MSCs来源EVs | 热应激提升MSC-EVs产量 | 提高MSC-EVs产量,增强药效 | [ | |
| 小鼠脂肪MSCs来源EVs | 缺氧提升MSC-EVs产量 | 同上 | [ | |
| 调控相关 机制通路 | 人胎盘MSCs来源EVs | 结肠特异性递送 | 抑制MAPK/NF-κB信号通路 | [ |
| 大鼠骨髓MSCs来源EVs | 高表达miR-146a的MSC-EVs | 调控NF-κB信号通路 | [ | |
| 减轻炎症反应 | 人脐带MSCs来源EVs | 高表达IL-27的MSC-EVs | 减少不良反应 | [ |
表 2 工程化MSC-EVs在IBD中的应用策略
Table 2 Strategies for the application of engineered MSC-EVs in IBD
| 策略 | EVs来源 | 特点 | 药效作用 | 参考文献 |
| 免疫调节 | 小鼠骨髓MSCs来源EVs | 高表达PD-L1的MSC-EVs | 重塑免疫微环境 | [ |
| 人牙周韧带MSCs来源EVs | 热应激提升MSC-EVs产量 | 增强免疫调节作用 | [ | |
| 增加药效与 递送效率 | 人脐带MSCs来源EVs | 包封MSC-EVs的微胶囊系统 | 提升MSC-EVs的稳定性、靶向性及药效 | [ |
| 人胎盘MSCs来源EVs | 结肠特异性递送 | 靶向结肠并可控释放MSC-EVs | [ | |
| 人胎盘MSCs来源EVs | MSC-EVs负载Ber | 提高药物溶解度和生物利用度 | [ | |
| 人骨髓MSCs来源EVs | MSC-EVs负载Wnt激动剂 | 增强药物积累和生物利用度 | [ | |
| 人牙周韧带MSCs来源EVs | 热应激提升MSC-EVs产量 | 提高MSC-EVs产量,增强药效 | [ | |
| 小鼠脂肪MSCs来源EVs | 缺氧提升MSC-EVs产量 | 同上 | [ | |
| 调控相关 机制通路 | 人胎盘MSCs来源EVs | 结肠特异性递送 | 抑制MAPK/NF-κB信号通路 | [ |
| 大鼠骨髓MSCs来源EVs | 高表达miR-146a的MSC-EVs | 调控NF-κB信号通路 | [ | |
| 减轻炎症反应 | 人脐带MSCs来源EVs | 高表达IL-27的MSC-EVs | 减少不良反应 | [ |
| 1 |
Liu P, Gao C, Chen H, et al. Receptor-mediated targeted drug delivery systems for treatment of inflammatory bowel disease: Opportunities and emerging strategies[J]. Acta Pharm Sin B, 2021, 11 (9): 2798- 2818.
doi: 10.1016/j.apsb.2020.11.003 |
| 2 | Ward D, Nyboe Andersen N, Gørtz S, et al. Tumor necrosis factor inhibitors in inflammatory bowel disease and risk of immune mediated inflammatory diseases [J]. Clin Gastroenterol Hepatol, 2024, 22(1): 135-143. e8. |
| 3 |
Yang Q, Li SS, Ou HB, et al. Exosome-based delivery strategies for tumor therapy: an update on modification, loading, and clinical application[J]. J Nanobiotechnol, 2024, 22 (1): 41.
doi: 10.1186/s12951-024-02298-7 |
| 4 |
Shan YL, Zhang MY, Tao EX, et al. Pharmacokinetic characteristics of mesenchymal stem cells in translational challenges[J]. Signal Transduction Targeted Therapy, 2024, 9 (1): 242.
doi: 10.1038/s41392-024-01936-8 |
| 5 |
Zhang Q, Shan Y, Shen L, et al. Renal remodeling by CXCL10-CXCR3 axis-recruited mesenchymal stem cells and subsequent IL4I1 secretion in lupus nephritis[J]. Signal Transduction Targeted Therapy, 2024, 9 (1): 325.
doi: 10.1038/s41392-024-02018-5 |
| 6 |
Ni Q, Zhen L, Zeng Z, et al. Mesenchymal stromal cells restrain the Th17 cell response via L-amino-acid oxidase within lymph nodes[J]. Cell Death Dis, 2024, 15 (9): 640.
doi: 10.1038/s41419-024-07024-7 |
| 7 |
Eggenhofer E, Benseler V, Kroemer A, et al. Mesenchymal stem cells are short-lived and do not migrate beyond the lungs after intravenous infusion[J]. Frontiers Immunol, 2012, 3, 297.
doi: 10.3389/fimmu.2012.00297 |
| 8 |
Li S, Li W, Wu X, et al. Immune cell-derived extracellular vesicles for precision therapy of inflammatory-related diseases[J]. J Control Release, 2024, 368, 533- 547.
doi: 10.1016/j.jconrel.2024.03.007 |
| 9 |
Kalluri R, Mcandrews KM. The role of extracellular vesicles in cancer[J]. Cell, 2023, 186 (8): 1610- 1626.
doi: 10.1016/j.cell.2023.03.010 |
| 10 |
Buzas EI. The roles of extracellular vesicles in the immune system[J]. Nat Rev Immunol, 2022, 23 (4): 236- 250.
doi: 10.1038/s41577-022-00763-8 |
| 11 |
Kalluri R. The biology and function of extracellular vesicles in immune response and immunity[J]. Immunity, 2024, 57 (8): 1752- 1768.
doi: 10.1016/j.immuni.2024.07.009 |
| 12 |
Das S, Lyon CJ, Hu T. A panorama of extracellular vesicle applications: from biomarker detection to therapeutics[J]. ACS Nano, 2024, 18 (14): 9784- 9797.
doi: 10.1021/acsnano.4c00666 |
| 13 |
Herrmann IK, Wood MJA, Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform[J]. Nat Nanotechnol, 2021, 16 (7): 748- 759.
doi: 10.1038/s41565-021-00931-2 |
| 14 |
Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles[J]. Ann Rev Cell Develop Biol, 2014, 30 (1): 255- 289.
doi: 10.1146/annurev-cellbio-101512-122326 |
| 15 |
刘苡佳, 刘斌, 胡奎, 等. 干细胞和巨噬细胞来源的外泌体对结肠炎的调控作用及其机制[J]. 实用医学杂志, 2025, 41 (3): 447- 453.
doi: 10.3969/j.issn.1006-5725.2025.03.022 |
| 16 |
Welsh JA, Goberdhan DCI, O'Driscoll L, et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches[J]. J Extracel Ves, 2024, 13 (2): e12404.
doi: 10.1002/jev2.12404 |
| 17 |
Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines[J]. J Extracel Ves, 2018, 7 (1): 1535750.
doi: 10.1080/20013078.2018.1535750 |
| 18 |
李青, 李博, 陈政升, 等. 团体标准《人多能干细胞来源的小细胞外囊泡》与《人间充质干细胞来源的小细胞外囊泡》解读[J]. 中国研究型医院, 2023, 10 (3): 47- 50.
doi: 10.19450/j.cnki.jcrh.2023.03.010 |
| 19 |
Chen M, Lin S, Zhou C, et al. From conventional to microfluidic: progress in extracellular vesicle separation and individual characterization[J]. Adv Healthc Mater, 2023, 12 (8): e2202437.
doi: 10.1002/adhm.202202437 |
| 20 |
Rupp AK, Rupp C, Keller S, et al. Loss of EpCAM expression in breast cancer derived serum exosomes: Role of proteolytic cleavage[J]. Gynecol Oncol, 2011, 122 (2): 437- 446.
doi: 10.1016/j.ygyno.2011.04.035 |
| 21 | Ma XX, Peng LW, Zhu XH, et al. Isolation, identification, and challenges of extracellular vesicles: emerging players in clinical applications[J]. Apoptosis, 2024, 30 (1/2): 422- 445. |
| 22 |
Hass R, Von Der Ohe J, Luo TJ. Human mesenchymal stroma/stem-like cell-derived taxol-loaded EVs/exosomes transfer anti-tumor microRNA signatures and express enhanced SDF-1-mediated tumor tropism[J]. Cell Commu Signal, 2024, 22 (1): 506.
doi: 10.1186/s12964-024-01886-2 |
| 23 |
Eldh M, Lötvall J, Malmhäll C, et al. Importance of RNA isolation methods for analysis of exosomal RNA: Evaluation of different methods[J]. Mol Immunol, 2012, 50 (4): 278- 286.
doi: 10.1016/j.molimm.2012.02.001 |
| 24 |
Xie M, Li CF, She Z, et al. Human umbilical cord mesenchymal stem cells derived extracellular vesicles regulate acquired immune response of lupus mouse in vitro[J]. Scientific Rep, 2022, 12 (1): 13101.
doi: 10.1038/s41598-022-17331-8 |
| 25 |
Zhang B, Lai RC, Sim WK, et al. Topical Application Of Mesenchymal Stem Cell Exosomes Alleviates The Imiquimod Induced Psoriasis-Like Inflammation[J]. Int J Mol Sci, 2021, 22 (2): 720.
doi: 10.3390/ijms22020720 |
| 26 | Qian W, Wu E, Chen H, et al. MSCs-exosomes can promote macrophage M2 polarization via exosomal miR-21-5p through Mesenteric injection: a promising way to attenuate murine colitis [J]. J Crohns Colitis, 2024: jjae110. |
| 27 |
Lee JH, Lötvall J, Cho BS. The anti-inflammatory effects of adipose tissue mesenchymal stem cell exosomes in a mouse model of inflammatory bowel disease[J]. Int J Mol Sci, 2023, 24 (23): 16877.
doi: 10.3390/ijms242316877 |
| 28 |
Zhang K, Guo J, Yan W, et al. Macrophage polarization in inflammatory bowel disease[J]. Cell Commun Signal, 2023, 21 (1): 367.
doi: 10.1186/s12964-023-01386-9 |
| 29 |
Cao L, Xu HX, Wang G, et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells attenuate dextran sodium sulfate-induced ulcerative colitis by promoting M2 macrophage polarization[J]. Int Immunopharmacol, 2019, 72, 264- 274.
doi: 10.1016/j.intimp.2019.04.020 |
| 30 |
Yang S, Liang X, Song J, et al. A novel therapeutic approach for inflammatory bowel disease by exosomes derived from human umbilical cord mesenchymal stem cells to repair intestinal barrier via TSG-6[J]. Stem Cell Res Ther, 2021, 12 (1): 315.
doi: 10.1186/s13287-021-02404-8 |
| 31 |
Liu H, Liang Z, Wang F, et al. Exosomes from mesenchymal stromal cells reduce murine colonic inflammation via a macrophage-dependent mechanism[J]. JCI Insight, 2019, 4 (24): e131273.
doi: 10.1172/jci.insight.131273 |
| 32 |
Geremia A, Biancheri P, Allan P, et al. Innate and adaptive immunity in inflammatory bowel disease[J]. Autoimmu Rev, 2014, 13 (1): 3- 10.
doi: 10.1016/j.autrev.2013.06.004 |
| 33 |
Chen QY, Duan XY, Xu M, et al. BMSC-EVs regulate Th17 cell differentiation in UC via H3K27me3[J]. Mol Immunol, 2020, 118, 191- 200.
doi: 10.1016/j.molimm.2019.12.019 |
| 34 |
Tian J, Zhu QG, Zhang YD, et al. Olfactory ecto-mesenchymal stem cell-derived exosomes ameliorate experimental colitis modulating Th1/Th17 and Treg cell responses[J]. Front Immunol, 2020, 11, 598322.
doi: 10.3389/fimmu.2020.598322 |
| 35 |
Citi S. Intestinal barriers protect against disease[J]. Science, 2018, 359 (6380): 1097- 1098.
doi: 10.1126/science.aat0835 |
| 36 |
Yu H, Yang X, Xiao X, et al. Human adipose mesenchymal stem cell-derived exosomes protect mice from DSS-induced inflammatory bowel disease by promoting intestinal-stem-cell and epithelial regeneration[J]. Aging Dis, 2021, 12 (6): 1423- 1437.
doi: 10.14336/AD.2021.0601 |
| 37 |
Liang X, Li C, Song J, et al. HucMSC-Exo promote mucosal healing in experimental colitis by accelerating intestinal stem cells and epithelium regeneration via Wnt signaling pathway[J]. Int J Nanomed, 2023, 18, 2799- 2818.
doi: 10.2147/IJN.S402179 |
| 38 |
Cai X, ZHang ZY, Yuan JT, et al. hucMSC-derived exosomes attenuate colitis by regulating macrophage pyroptosis via the miR-378a-5p/NLRP3 axis[J]. Stem Cell Res Ther, 2021, 12 (1): 416.
doi: 10.1186/s13287-021-02492-6 |
| 39 |
Heidari N, Abbasi-Kenarsari H, Namaki S, et al. Adipose-derived mesenchymal stem cell-secreted exosome alleviates dextran sulfate sodium-induced acute colitis by Treg cell induction and inflammatory cytokine reduction[J]. J Cell Physiol, 2021, 236 (8): 5906- 5920.
doi: 10.1002/jcp.30275 |
| 40 |
Wei Z, Hang S, Wiredu Ocansey DK, et al. Human umbilical cord mesenchymal stem cells derived exosome shuttling mir-129-5p attenuates inflammatory bowel disease by inhibiting ferroptosis[J]. J Nanobiotechnol, 2023, 21 (1): 188.
doi: 10.1186/s12951-023-01951-x |
| 41 |
Yang R, Huang H, Cui S, et al. IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b[J]. Cell Death Dis, 2020, 11 (7): 603.
doi: 10.1038/s41419-020-02788-0 |
| 42 |
Wong WY, Lee MML, Chan BD, et al. Proteomic profiling of dextran sulfate sodium induced acute ulcerative colitis mice serum exosomes and their immunomodulatory impact on macrophages[J]. Proteomics, 2016, 16 (7): 1131- 1145.
doi: 10.1002/pmic.201500174 |
| 43 | Xu F, Fei Z, Dai H, et al. Mesenchymal stem cell‐derived extracellular vesicles with high PD‐L1 expression for autoimmune diseases treatment[J]. Adv Materials, 2021, 34 (1): e2106265. |
| 44 |
Nie M, Huang D, Chen G, et al. Bioadhesive microcarriers encapsulated with IL‐27 high expressive MSC extracellular vesicles for inflammatory bowel disease treatment[J]. Advanced Science, 2023, 10 (32): e2303349.
doi: 10.1002/advs.202303349 |
| 45 |
Wu H, Fan H, Shou Z, et al. Extracellular vesicles containing miR-146a attenuate experimental colitis by targeting TRAF6 and IRAK1[J]. Int Immunopharmacol, 2019, 68, 204- 212.
doi: 10.1016/j.intimp.2018.12.043 |
| 46 |
Ambattu LA, Ramesan S, Dekiwadia C, et al. High frequency acoustic cell stimulation promotes exosome generation regulated by a calcium-dependent mechanism[J]. Commun Biol, 2020, 3 (1): 553.
doi: 10.1038/s42003-020-01277-6 |
| 47 |
Tang D, Liu M, Gao S, et al. Thermally engineered MSC-derived extracellular vesicles ameliorate colitis in mice by restoring the imbalanced Th17/Treg cell ratio[J]. Int Immunopharmacoly, 2023, 125, 111077.
doi: 10.1016/j.intimp.2023.111077 |
| 48 |
Qian W, Huang L, Xu Y, et al. Hypoxic ASCs-derived Exosomes attenuate colitis by regulating macrophage polarization via miR-216a-5p/HMGB1 axis[J]. Inflamma Bowel Dis, 2023, 29 (4): 602- 619.
doi: 10.1093/ibd/izac225 |
| 49 |
Deng C, Hu Y, Conceição M, et al. Oral delivery of layer-by-layer coated exosomes for colitis therapy[J]. J Control Rel, 2023, 354, 635- 650.
doi: 10.1016/j.jconrel.2023.01.017 |
| 50 |
Gan J, Sun L, Chen G, et al. Mesenchymal stem cell exosomes encapsulated oral microcapsules for acute colitis treatment[J]. Advanced Healthcare Materials, 2022, 11 (17): e2201105.
doi: 10.1002/adhm.202201105 |
| 51 |
Mofazali P, Atapour M, Nakamura M, et al. Evaluation of layer-by-layer assembly systems for drug delivery and antimicrobial properties in orthopaedic application[J]. Int J Pharmac, 2024, 657, 124148.
doi: 10.1016/j.ijpharm.2024.124148 |
| 52 |
Borges J, Zeng J, Liu XQ, et al. Recent developments in layer‐by‐layer assembly for drug delivery and tissue engineering applications[J]. Advanced Healthcare Materials, 2024, 13 (8): e2302713.
doi: 10.1002/adhm.202302713 |
| 53 |
Barile L, Vassalli G. Exosomes: Therapy delivery tools and biomarkers of diseases[J]. Pharmacol Ther, 2017, 174, 63- 78.
doi: 10.1016/j.pharmthera.2017.02.020 |
| 54 |
Deng C, Hu Y, Conceicao M, et al. Oral delivery of layer-by-layer coated exosomes for colitis therapy[J]. J Control Release, 2023, 354, 635- 650.
doi: 10.1016/j.jconrel.2023.01.017 |
| 55 |
Tang X, Shang Y, Yang H, et al. Targeted delivery of Fc-fused PD-L1 for effective management of acute and chronic colitis[J]. Nat Commu, 2024, 15 (1): 1673.
doi: 10.1038/s41467-024-46025-0 |
| 56 |
Fu YJ, Zhao X, Wang LY, et al. A gas therapy strategy for intestinal flora regulation and colitis treatment by nanogel‐based multistage NO delivery microcapsules[J]. Advanced Materials, 2024, 36 (19): e2309972.
doi: 10.1002/adma.202309972 |
| 57 |
Zhang Y, Ma R, You C, et al. Hyaluronic acid modified oral drug delivery system with mucoadhesiveness and macrophage-targeting for colitis treatment[J]. Carbohydrate Polymers, 2023, 313, 120884.
doi: 10.1016/j.carbpol.2023.120884 |
| 58 |
王心怡, 付淑军, 孟祥骏, 等. 纳米药物递送系统体内命运分析新方法与新技术研究进展[J]. 药学进展, 2024, 48 (10): 747- 760.
doi: 10.20053/j.issn1001-5094.2024.10.004 |
| 59 |
Sun L, Liu H, Ye Y, et al. Smart nanoparticles for cancer therapy[J]. Signal Transduction Targeted Therapy, 2023, 8 (1): 418.
doi: 10.1038/s41392-023-01642-x |
| 60 |
Zhao S, Di Y, Fan H, et al. Targeted delivery of extracellular vesicles: the mechanisms, techniques and therapeutic applications[J]. Mol Biomed, 2024, 5 (1): 60.
doi: 10.1186/s43556-024-00230-x |
| 61 |
Deng C, Zhang H, Li Y, et al. Exosomes derived from mesenchymal stem cells containing berberine for ulcerative colitis therapy[J]. J Coll Inter Sci, 2024, 671, 354- 373.
doi: 10.1016/j.jcis.2024.05.162 |
| 62 |
Guo J, Wang F, Hu Y, et al. Exosome-based bone-targeting drug delivery alleviates impaired osteoblastic bone formation and bone loss in inflammatory bowel diseases[J]. Cell Rep Med, 2023, 4 (1): 100881.
doi: 10.1016/j.xcrm.2022.100881 |
| 63 |
Greenberg ZF, Graim KS, He M. Towards artificial intelligence-enabled extracellular vesicle precision drug delivery[J]. Advanced Drug Delivery Reviews, 2023, 199, 114974.
doi: 10.1016/j.addr.2023.114974 |
| 64 |
Nademi Y, Tang T, Uludağ H. Modeling uptake of polyethylenimine/short interfering RNA nanoparticles in breast cancer cells using machine learning[J]. Advanced NanoBiomed Res, 2021, 1 (10): 2000106.
doi: 10.1002/anbr.202000106 |
| [1] | 王倩, 梁昌昌, 沈士朋, 刘茂东, 白璐. IL-33/ST2信号通路在肾脏疾病中作用机制的研究进展[J]. 中国临床药理学与治疗学, 2025, 30(8): 1122-1126. |
| [2] | 周莎莎, 程雪清, 彭冬冬, 王小青, 扶丽君, 肖文喜, 张国民. 清热消炎宁抗甲型流感H3N2病毒的药效及作用机制研究[J]. 中国临床药理学与治疗学, 2025, 30(3): 347-354. |
| [3] | 董家珊, 陈嘉睿, 曾大勇, 刘亦伟, 许建文, 林荣芳. 炎症性肠病患者GST基因多态性与硫唑嘌呤活性代谢物6-TGN浓度水平相关性研究[J]. 中国临床药理学与治疗学, 2025, 30(10): 1383-1390. |
| [4] | 刘丽桦, 刘德伍. 脂肪间充质干细胞外泌体源非编码RNA在创面愈合中的作用[J]. 中国临床药理学与治疗学, 2024, 29(9): 1049-1056. |
| [5] | 李培培, 武岳, 张贤政, 张玲玲. 维得利珠单抗治疗炎症性肠病患者疗效评价的回顾性研究[J]. 中国临床药理学与治疗学, 2024, 29(8): 899-906. |
| [6] | 赵婷婷, 李俊峰, 张立婷. 间充质干细胞源性外泌体对肝纤维化潜在治疗机制的研究进展[J]. 中国临床药理学与治疗学, 2024, 29(4): 475-480. |
| [7] | 于淼瑛, 王艳辉, 阎石. 中药活性成分干预骨髓间充质干细胞衰老分子机制的研究进展[J]. 中国临床药理学与治疗学, 2024, 29(1): 99-106. |
| [8] | 许金回, 岳红梅, 李雅亭, 刘苗苗, 武兴东, 朱浩斌. 间充质干细胞对OSAHS病理生理调节机制的研究进展[J]. 中国临床药理学与治疗学, 2024, 29(1): 114-120. |
| [9] | 余伟迪, 宋振顺. 骨髓间充质干细胞通过抑制铁死亡改善大鼠重症急性胰腺炎相关肺损伤[J]. 中国临床药理学与治疗学, 2022, 27(7): 721-728. |
| [10] | 曹颖. 自噬与炎症性肠病关系的研究进展[J]. 中国临床药理学与治疗学, 2022, 27(3): 345-352. |
| [11] | 邓颖, 白殊同. 金圣草黄素相关药理作用机制的研究进展 [J]. 中国临床药理学与治疗学, 2022, 27(10): 1155-1162. |
| [12] | 段睿潇, 赵一帆, 叶永娟, 刘 敏, 李 强, 周永宁. 胆汁酸代谢调节炎症性肠病的作用机制及药物研发[J]. 中国临床药理学与治疗学, 2022, 27(10): 1171-1181. |
| [13] | 彭静, 宋静, 栾家杰. 新型冠状病毒肺炎治疗新策略:间充质干细胞疗法[J]. 中国临床药理学与治疗学, 2021, 26(9): 1073-1079. |
| [14] | 章权, 石仕元, 韩贵和. 1,25二羟维生素D3在脊柱结核治疗中的免疫调节作用[J]. 中国临床药理学与治疗学, 2021, 26(7): 787-793. |
| [15] | 路明, 尹晓玉, 张志清. 间充质干细胞对自身免疫病治疗作用研究进展及机制探究[J]. 中国临床药理学与治疗学, 2021, 26(6): 700-706. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||