中国临床药理学与治疗学 ›› 2026, Vol. 31 ›› Issue (3): 313-323.doi: 10.12092/j.issn.1009-2501.2026.03.003
刘晓绵1( ), 刘颖1, 涂文婧1, 张硕1, 朱高彦1, 崔语嫣1, 崔肖雨1, 杨一璇1, 李晓冰2,*(
), 刘颖1, 涂文婧1, 张硕1, 朱高彦1, 崔语嫣1, 崔肖雨1, 杨一璇1, 李晓冰2,*( ), 郭洪涛3, 李栋4, 何小鹃5
), 郭洪涛3, 李栋4, 何小鹃5
收稿日期:2025-05-09
修回日期:2025-08-28
出版日期:2026-03-26
发布日期:2026-04-03
通讯作者:
李晓冰
E-mail:lxmianjyjy@163.com;baishaoyao@163.com
作者简介:刘晓绵,女,研究方向:中西医结合防治风湿免疫病。E-mail:基金资助:
Xiaomian LIU1( ), Ying LIU1, Wenjing TU1, Shuo ZHANG1, Gaoyan ZHU1, Yuyan CUI1, Xiaoyu CUI1, Yixuan YANG1, Xiaobing LI2,*(
), Ying LIU1, Wenjing TU1, Shuo ZHANG1, Gaoyan ZHU1, Yuyan CUI1, Xiaoyu CUI1, Yixuan YANG1, Xiaobing LI2,*( ), Hongtao GUO3, Dong LI4, Xiaojuan HE5
), Hongtao GUO3, Dong LI4, Xiaojuan HE5
Received:2025-05-09
Revised:2025-08-28
Online:2026-03-26
Published:2026-04-03
Contact:
Xiaobing LI
E-mail:lxmianjyjy@163.com;baishaoyao@163.com
摘要:
目的: 探讨加味二妙散(modified Er Miao-San,MEMS)治疗急性痛风性关节炎(acute gouty arthritis,AGA)的疗效及潜在作用机制。方法: 通过网络药理学、分子对接和分子动力学模拟验证MEMS活性成分与AGA靶点间的相互作用。通过向大鼠右踝关节注射25 mg/kg尿酸钠(MSU)悬液建立AGA模型。通过测量踝关节肿胀度、步态评分和炎症指数评估治疗效果;ELISA法测定AGA大鼠血清TNF-α、IL-6、IL-1β、IL-17、MDA、SOD和GSH浓度;HE染色观察踝关节滑膜病理变化;Western blot分析各组踝关节滑膜NLRP3和STAT3蛋白水平。结果: 网络药理学分析发现,槲皮素、β-谷甾醇、豆甾醇和汉黄芩素是MEMS治疗AGA的核心活性成分。TNF、STAT3、IL-6和IL-1β是MEMS治疗AGA的关键靶点,核心活性成分与关键靶点具有较强的结合活性和稳定构象。关键靶点主要富集于NOD样受体、IL-17和TNF等信号通路。动物模型研究表明,MEMS能有效减轻AGA大鼠踝关节肿胀度,改善步态评分和炎症指数,减轻踝关节滑膜细胞增殖、中性粒细胞浸润和毛细血管充血,降低血清TNF-α、IL-1β、IL-6、IL-17和MDA水平,提高SOD和GSH含量,下调踝关节滑膜NLRP3和STAT3蛋白表达。结论: MEMS能减轻AGA大鼠炎症和氧化应激损伤,发挥抗炎抗氧化功能,其作用机制可能与通过抑制STAT3/NLRP3信号通路调控炎症因子水平有关。
中图分类号:
刘晓绵, 刘颖, 涂文婧, 张硕, 朱高彦, 崔语嫣, 崔肖雨, 杨一璇, 李晓冰, 郭洪涛, 李栋, 何小鹃. 网络药理学与分子对接联合实验验证的加味二妙散抗急性痛风性关节炎机制研究[J]. 中国临床药理学与治疗学, 2026, 31(3): 313-323.
Xiaomian LIU, Ying LIU, Wenjing TU, Shuo ZHANG, Gaoyan ZHU, Yuyan CUI, Xiaoyu CUI, Yixuan YANG, Xiaobing LI, Hongtao GUO, Dong LI, Xiaojuan HE. Unraveling the molecular mechanisms of modified Er-Miao-San (MEMS) in acute gouty arthritis: a multidisciplinary integration of network pharmacology, molecular docking, and experimental validation[J]. Chinese Journal of Clinical Pharmacology and Therapeutics, 2026, 31(3): 313-323.

图 3
Fig.3 The TCM-active components-drug targets network Rectangles represent the intersection targets of drugs and diseases, triangles represent the core active components of drugs, rhombuses represent the categories of traditional Chinese medicine, and concave quadrilaterals represent the common active components of drugs.
| Number | Molecule ID | Molecule name | OB(%) | DL | TCM | Degree |
| 1 | MOL000098 | Quercetin | 46.43 | 0.28 | HuangBai, TuFuLing | 51 |
| 2 | MOL000358 | beta-sitosterol | 36.91 | 0.75 | HuangBai, TuFuLing, ShanCiGu | 37 |
| 3 | MOL000173 | wogonin | 30.68 | 0.23 | CangZhu | 29 |
| 4 | MOL000449 | Stigmasterol | 43.83 | 0.76 | HuangBai, TuFuLing, ShanCiGu | 29 |
表 1
Table 1 Core active compounds of MEMS and their degree values
| Number | Molecule ID | Molecule name | OB(%) | DL | TCM | Degree |
| 1 | MOL000098 | Quercetin | 46.43 | 0.28 | HuangBai, TuFuLing | 51 |
| 2 | MOL000358 | beta-sitosterol | 36.91 | 0.75 | HuangBai, TuFuLing, ShanCiGu | 37 |
| 3 | MOL000173 | wogonin | 30.68 | 0.23 | CangZhu | 29 |
| 4 | MOL000449 | Stigmasterol | 43.83 | 0.76 | HuangBai, TuFuLing, ShanCiGu | 29 |
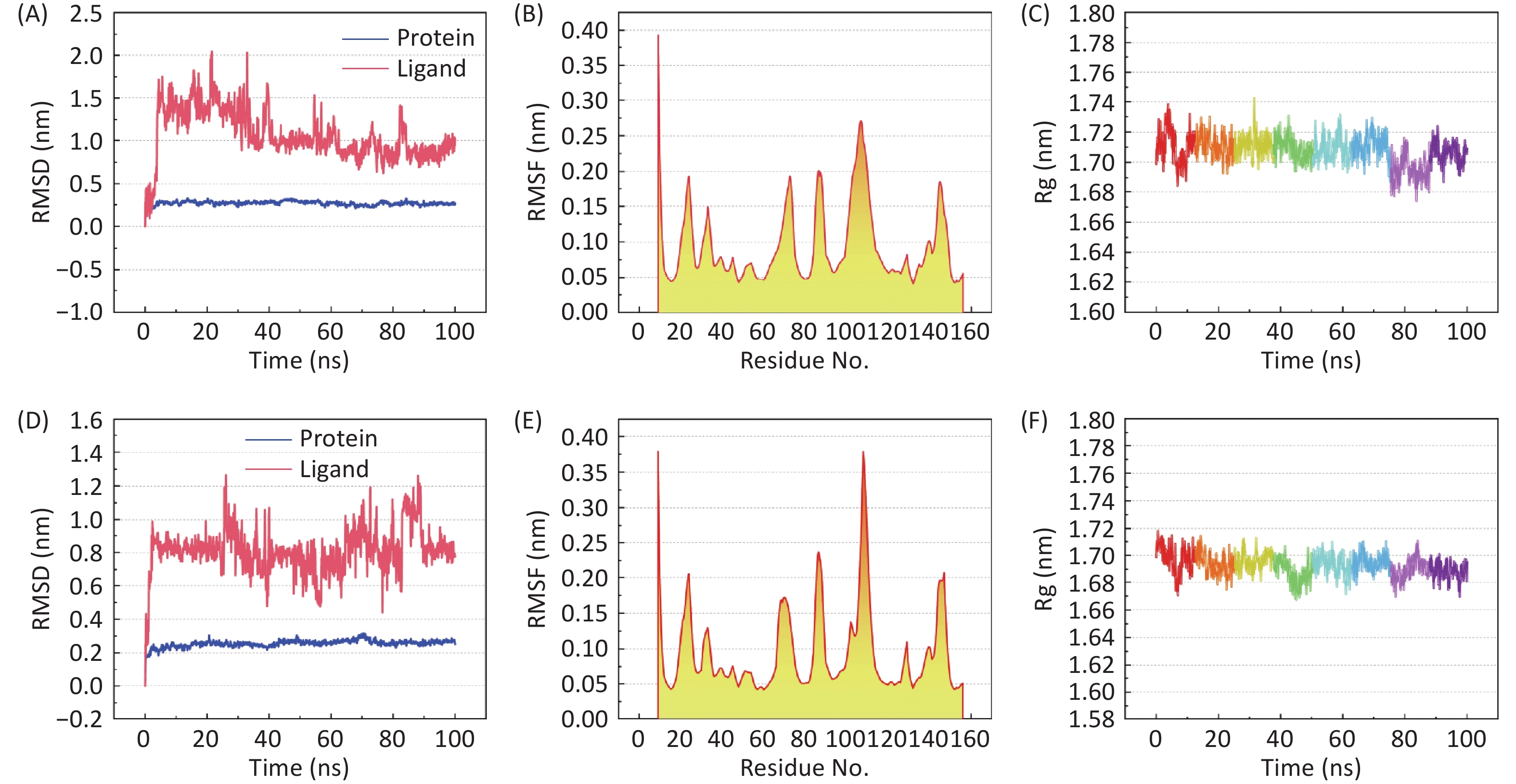
图 8
Fig.8 Conformational stability analysis of TNF in complex with β-sitosterol or stigmasterol A-C: RMSD, RMSF, and Rg profiles of the TNF-β-sitosterol complex; D-F: corresponding profiles of the TNF-stigmasterol complex.
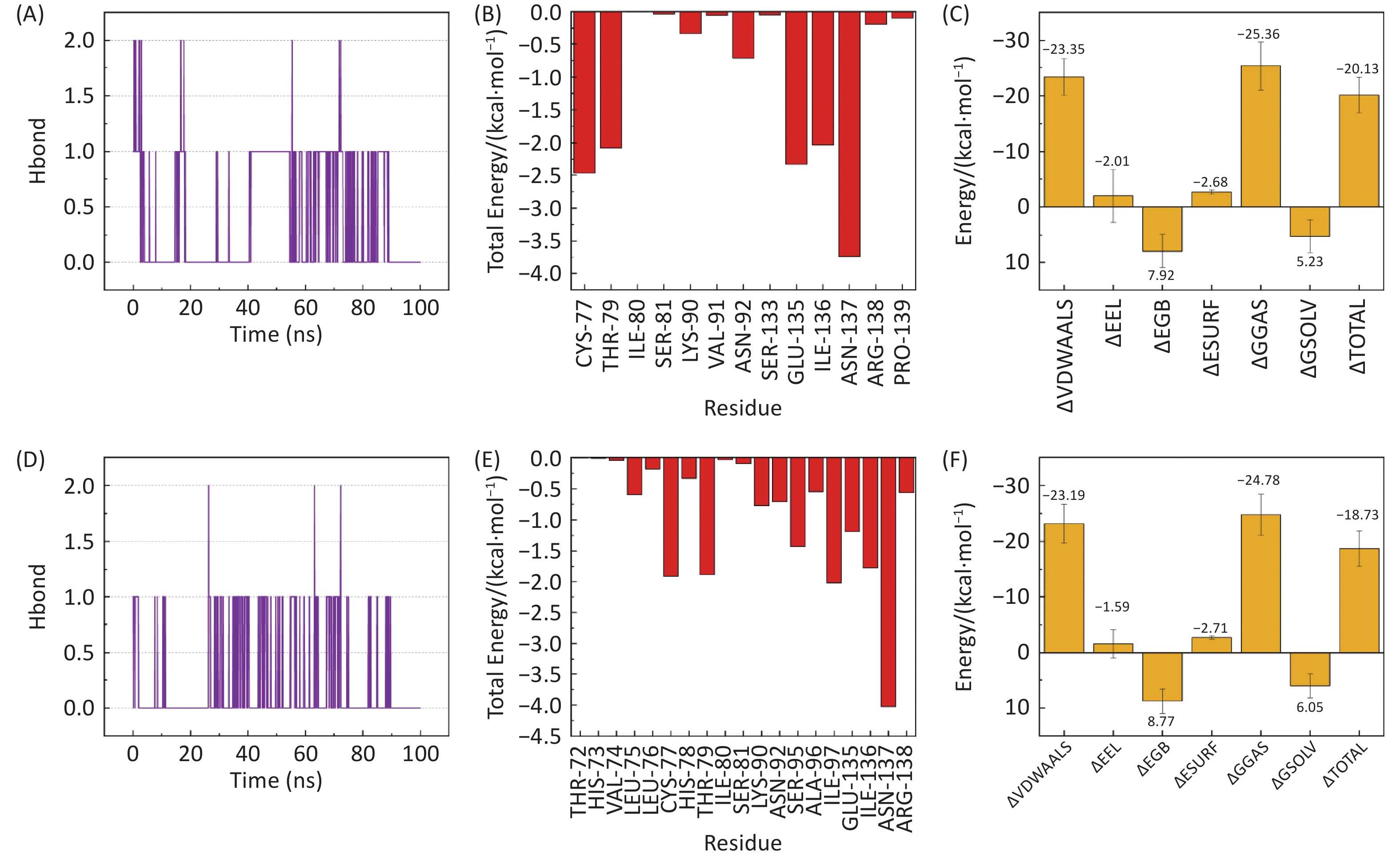
图 9
Fig.9 Hydrogen bonding and binding free energy analysis A,B: number of hydrogen bonds in the TNF-β-sitosterol and TNF-stigmasterol complexes, respectively. C,D: binding free energy decomposition. E,F: per-residue energy contributions for each complex.

图 11
Fig.11 Effects of MEMS on ankle joint swelling, toe volume swelling, gait score, and inflammatory index in AGA rats A: ankle swelling before and 12 h after MSU injection; B: comparison of the degree of right posterior ankle joint swelling, the degree of toe volume ankle swelling, gait assessment score and Inflammatory index in rats of different group ($ \overline{x} $±s, n=6). bP<0.05, cP<0.01, compared with model group; eP<0.05, compared with EMS group.
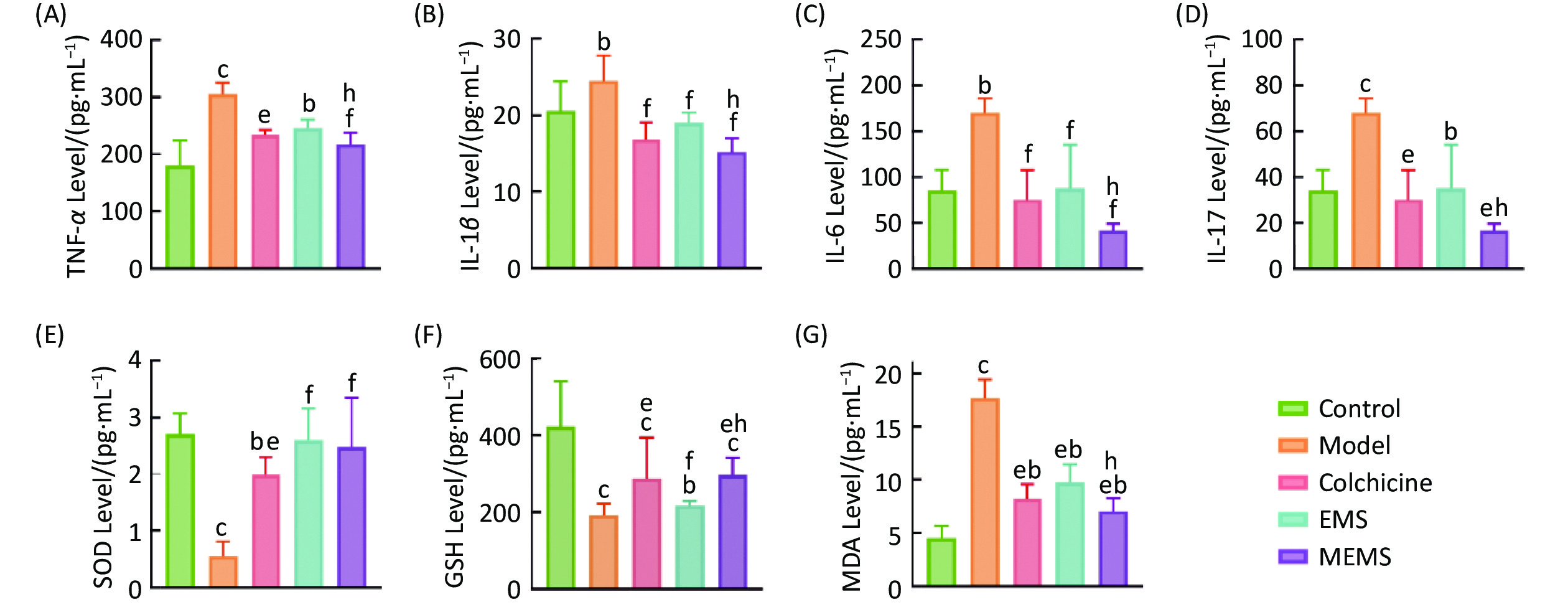
图 12
Fig.12 Results of various indexes in rats ($ \overline{x} $±s, n=6) A: TNF-α; B: IL-1β; C: IL-6; D: IL-17; E: SOD; F: GSH; G: MDA. bP<0.05, cP<0.01, compared with control group; eP<0.05, fP<0.01, compared with Model group; hP<0.05, compared with EMS group.
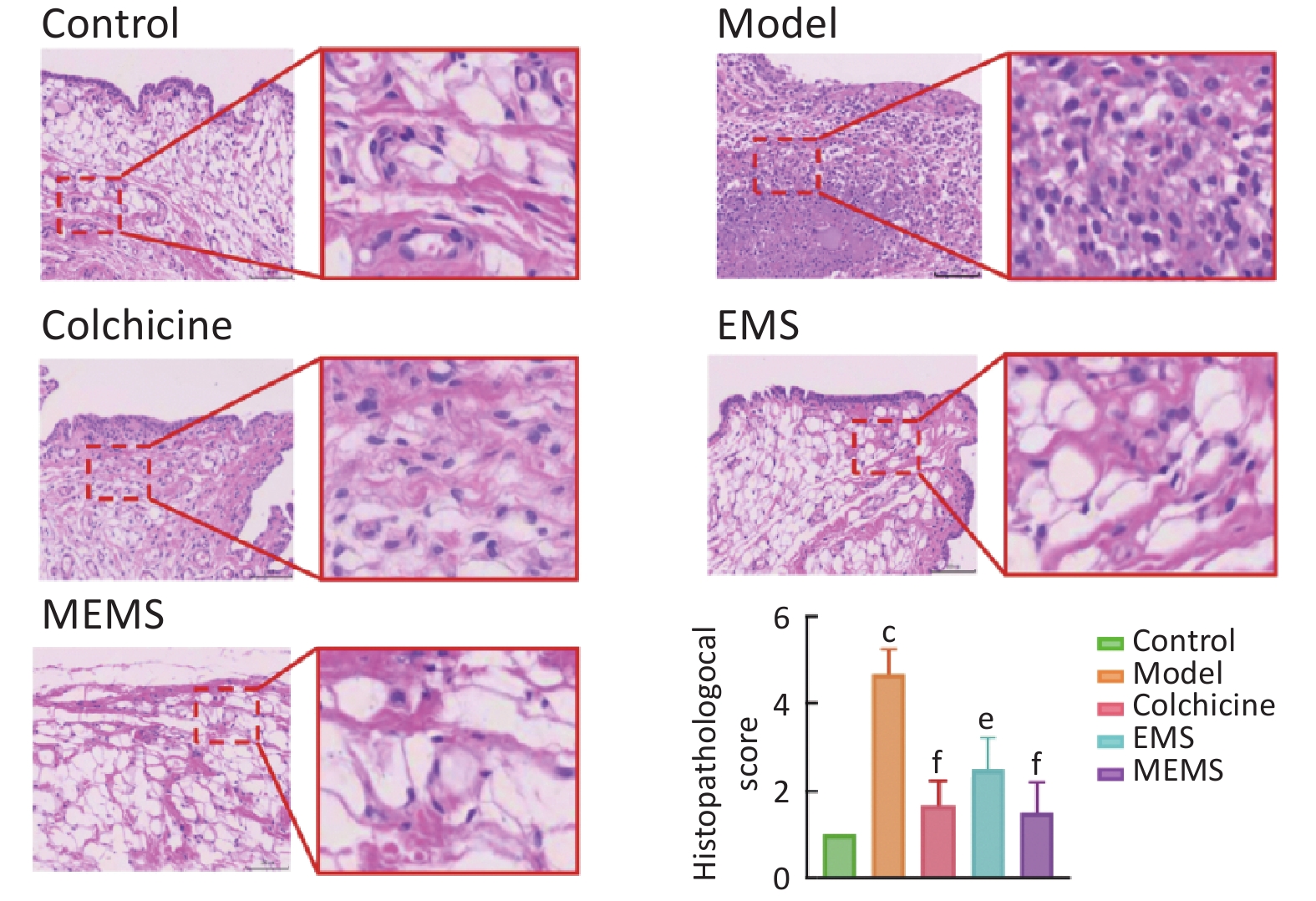
图 13
Fig.13 HE staining of synovial tissues in rat ankle joints across experimental groups ($ \overline{x} $±s, n=6) The representative image of HE staining in synovial tissue (Scale bar=100 μm); the histological quantitative score of synovial tissue sections was average±SD. cP<0.01, compared with control group; eP<0.05, fP<0.01, compared with model group.
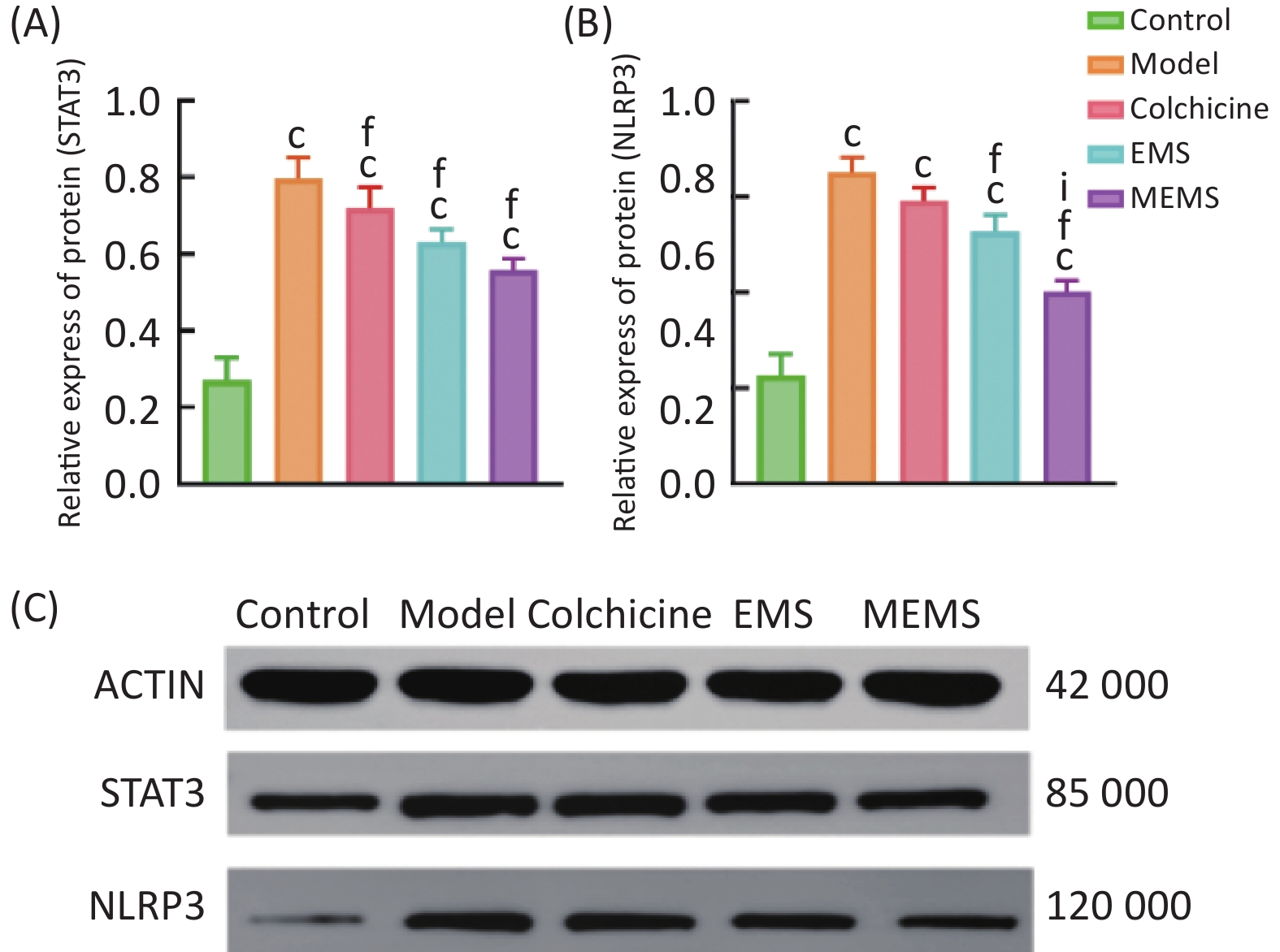
图 14
Fig.14 The expression levels of NLRP3 and STAT3 protein in ankle joint tissue of rats ($ \overline{x} $±s, n=6) A: STAT3 protein expression; B: NLRP3 protein expression; C: electrophoresis of STAT3 and NLRP3 proteins. cP<0.01, compared with control group; fP<0.01, compared with model group; iP<0.01, compared with EMS group.
| 1 |
Patil T, Soni A, Acharya S. A brief review on in vivo models for Gouty Arthritis[J]. Metab Open, 2021, 11, 100100.
doi: 10.1016/j.metop.2021.100100 |
| 2 |
Cross M, Ong KL, Culbreth GT, et al. Global, regional, and national burden of gout, 1990–2020, and projections to 2050: a systematic analysis of the Global Burden of Disease Study 2021[J]. Lancet Rheumatol, 2024, 6 (8): e507- e517.
doi: 10.1016/S2665-9913(24)00117-6 |
| 3 |
Guo B, Zhao CP, Zhang C, et al. Elucidation of the anti-inflammatory mechanism of Er Miao San by integrative approach of network pharmacology and experimental verification[J]. Pharmacol Res, 2022, 175, 106000.
doi: 10.1016/j.phrs.2021.106000 |
| 4 | Chen G, Li KK, Fung CH, et al. Er-Miao-San, a traditional herbal formula containing Rhizoma Atractylodis and Cortex Phellodendri inhibits inflammatory mediators in LPS-stimulated RAW264. 7 macrophages through inhibition of NF-κB pathway and MAPKs activation [J]. J Ethnopharmacol, 2014, 154(3): 711-718. |
| 5 |
梁浩瀚, 崔伟, 叶来生, 等. 土茯苓及其活性成分防治痛风性关节炎作用与机制研究进展[J]. 中药材, 2023, 46 (10): 2628- 2639.
doi: 10.13863/j.issn1001-4454.2023.10.042 |
| 6 |
陈诗, 赵玥, 王振, 等. 山慈菇药理作用及临床应用研究进展[J]. 中华中医药学刊, 2023, 41 (6): 247- 250.
doi: 10.13193/j.issn.1673-7717.2023.06.054 |
| 7 |
袁莉莉, 王倩. 基于网络药理学研究泽泻汤治疗脑水肿的作用机制[J]. 长春中医药大学学报, 2019, 35 (5): 912- 915.
doi: 10.13463/j.cnki.cczyy.2019.05.028 |
| 8 |
王芳, 李峰. 基于网络药理学研究二妙方治疗新型冠状病毒肺炎的潜在作用机制[J]. 现代中药研究与实践, 2022, 36 (3): 43- 48.
doi: 10.13728/j.1673-6427.2022.03.009 |
| 9 |
Coderre TJ, Wall PD. Ankle joint urate arthritis (AJUA) in rats: an alternative animal model of arthritis to that produced by Freund's adjuvant[J]. Pain, 1987, 28 (3): 379- 393.
doi: 10.1016/0304-3959(87)90072-8 |
| 10 |
Li H, Zhang X, Gu L, et al. Anti-gout effects of the medicinal fungus phellinus igniarius in hyperuricaemia and acute gouty arthritis rat models[J]. Front Pharmacol, 2022, 12, 801910.
doi: 10.3389/fphar.2021.801910 |
| 11 |
肖敬, 尹智功, 蒋耀平, 等. 青藤碱对痛风性关节炎模型大鼠IL-6的影响[J]. 河南中医, 2018, 38 (4): 536- 539.
doi: 10.16367/j.issn.1003-5028.2018.04.0143 |
| 12 |
陈海娟, 高禹涵, 徐丽, 等. 十味乳香胶囊对类风湿性关节炎大鼠的治疗作用[J]. 华西药学杂志, 2024, 39 (3): 274- 278.
doi: 10.13375/j.cnki.wcjps.2024.03.007 |
| 13 |
王赓丰, 马俊福, 邓雍钲, 等. 基于NF-κB通路探讨痛风汤对急性痛风性关节炎模型大鼠炎症的影响机制[J]. 陕西中医, 2024, 45 (6): 740- 744.
doi: 10.3969/j.issn.1000-7369.2024.06.004 |
| 14 |
王海坤, 苏丹, 吴娜, 等. 基于网络药理学和分子对接探讨上中下通用痛风方治疗痛风性关节炎的分子机制[J]. 特产研究, 2023, 45 (2): 66- 73+80.
doi: 10.16720/j.cnki.tcyj.2023.043 |
| 15 |
de Sá Müller CM, Coelho GB, Araújo MCPM, et al. Lychnophora pinaster ethanolic extract and its chemical constituents ameliorate hyperuricemia and related inflammation[J]. J Ethnopharmacol, 2019, 242, 112040.
doi: 10.1016/j.jep.2019.112040 |
| 16 |
Liu F, Shen F, Bai Y, et al. Mechanism of DaiTongXiao in the treatment of gouty arthritis through the NLRP3 signaling pathway[J]. J Ethnopharmacol, 2024, 319, 117313.
doi: 10.1016/j.jep.2023.117313 |
| 17 |
Kobayashi EH, Suzuki T, Funayama R, et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription[J]. Nat Commun, 2016, 7 (1): 11624.
doi: 10.1038/ncomms11624 |
| 18 |
Lin G, Yu Q, Xu L, et al. Berberrubine attenuates potassium oxonate-and hypoxanthine-induced hyperuricemia by regulating urate transporters and JAK2/STAT3 signaling pathway[J]. Eur J Pharmacol, 2021, 912, 174592.
doi: 10.1016/j.ejphar.2021.174592 |
| 19 |
Wu M, Tian Y, Wang Q, et al. Gout: a disease involved with complicated immunoinflammatory responses: a narrative review[J]. Clin Rheumatol, 2020, 39 (10): 2849- 2859.
doi: 10.1007/s10067-020-05090-8 |
| 20 |
Lin Y, Luo T, Weng A, et al. Gallic acid alleviates gouty arthritis by inhibiting NLRP3 inflammasome activation and pyroptosis through enhancing Nrf2 signaling[J]. Front Immunol, 2020, 11, 580593.
doi: 10.3389/fimmu.2020.580593 |
| 21 |
Luo L, Wang F, Xu X, et al. STAT3 promotes NLRP3 inflammasome activation by mediating NLRP3 mitochondrial translocation[J]. Exp Mol Med, 2024, 56 (9): 1980- 1990.
doi: 10.1038/s12276-024-01298-9 |
| 22 |
Reber LL, Marichal T, Sokolove J, et al. Contribution of mast cell-derived interleukin‐1β to uric acid crystal-induced acute arthritis in mice[J]. Arthritis Rheumatol, 2014, 66 (10): 2881- 2891.
doi: 10.1002/art.38747 |
| 23 |
Lv Y, Qi J, Babon JJ, et al. The JAK-STAT pathway: from structural biology to cytokine engineering[J]. Signal Transduct Target Ther, 2024, 9 (1): 221.
doi: 10.1038/s41392-024-01934-w |
| 24 |
Jiang Z, Yin X, Wang M, et al. β-Hydroxybutyrate alleviates pyroptosis in MPP+/MPTP-induced Parkinson’s disease models via inhibiting STAT3/NLRP3/GSDMD pathway[J]. Int Immunopharmacol, 2022, 113, 109451.
doi: 10.1016/j.intimp.2022.109451 |
| 25 |
Mariotte A, De Cauwer A, Po C, et al. A mouse model of MSU-induced acute inflammation in vivo suggests imiquimod-dependent targeting of Il-1β as relevant therapy for gout patients[J]. Theranostics, 2020, 10 (5): 2158.
doi: 10.7150/thno.40650 |
| 26 |
Martinon F, Pétrilli V, Mayor A, et al. Gout-associated uric acid crystals activate the NALP3 inflammasome[J]. Nature, 2006, 440 (7081): 237- 241.
doi: 10.1038/nature04516 |
| 27 |
Xu X, Yu D, Wang Y, et al. Investigating the mechanisms of resveratrol in the treatment of gouty arthritis through the integration of network pharmacology and metabolics[J]. Front Endocrinol, 2024, 15, 1438405.
doi: 10.3389/fendo.2024.1438405 |
| 28 |
Hao K, Jiang W, Zhou M, et al. Targeting BRD4 prevents acute gouty arthritis by regulating pyroptosis[J]. Int J Biol Sci, 2020, 16 (16): 3163.
doi: 10.7150/ijbs.46153 |
| [1] | 温子帅, 梁胜男, 阮雨玲, 张文涛, 李梦颖, 吴芳芳, 柳俊辉, 秦华珍. 基于网络药理学、分子对接及实验验证探讨高良姜等三味山姜属中药治疗胃溃疡寒证的作用机制[J]. 中国临床药理学与治疗学, 2026, 31(1): 28-39. |
| [2] | 李倩, 王贞香, 梁艳婷, 马玮玮, 张振, 王霞, 安琼. 基于网络药理学、分子对接和动物实验探究西河柳对链脲佐菌素诱导的糖尿病大鼠的影响及作用机制[J]. 中国临床药理学与治疗学, 2025, 30(7): 907-920. |
| [3] | 郭玲, 彭小勇, 邓蒙生, 朱英国, 翁昌梅, 程祥云, 王建民, 李涛, 刘良明, 杨光明. 瞬时受体电位香草酸亚型1激动剂辣椒素对创伤失血休克大鼠的保护作用[J]. 中国临床药理学与治疗学, 2025, 30(6): 721-731. |
| [4] | 杨卫东, 王瑞麒, 王海花, 叶田香, 程生辉, 李会芳, 郝旭亮. 基于网络药理学、分子对接及动物实验探讨毛建茶黄酮提取物改善痛风性关节炎的作用机制[J]. 中国临床药理学与治疗学, 2025, 30(6): 763-773. |
| [5] | 李明奇, 王映荷, 赵晓璐, 包小妹, 岳鑫, 任贵强, 马月宏. 基于LC-MS/MS结合网络药理学、分子对接及体内外实验探究红花总黄酮抗肝纤维化的作用机制[J]. 中国临床药理学与治疗学, 2025, 30(5): 586-598. |
| [6] | 刘艮, 杨卫东, 李佳, 刘聪, 郝旭亮. 基于网络药理学探讨白连降浊方治疗脂质异常的机理[J]. 中国临床药理学与治疗学, 2025, 30(4): 464-476. |
| [7] | 张慧慧, 金乐, 刘苏, 陈洪晓, 陈昭琳, 唐丽琴. 小檗碱抑制乳腺癌细胞增殖的网络药理学分析及体外细胞验证[J]. 中国临床药理学与治疗学, 2025, 30(3): 332-338. |
| [8] | 付晓艳, 巩子汉, 高广淼, 杨必乾, 邓毅, 王立平, 杨秀娟, 杨志军. 基于网络药理学、分子对接和动物实验探讨甘草缓解马钱子肝损伤的作用机制[J]. 中国临床药理学与治疗学, 2025, 30(10): 1326-1341. |
| [9] | 雷程静, 于淼, 李艳格, 唐小光, 赵繁荣, 祝田田. 基于网络药理学及动物实验探讨心脉佳制剂抑制高血压心肌肥大的作用机制[J]. 中国临床药理学与治疗学, 2025, 30(1): 32-41. |
| [10] | 策力木格, 胡德力根, 许良. 网络药理学-分子对接分析及实验验证蒙药高尤的肾功能保护作用机制[J]. 中国临床药理学与治疗学, 2024, 29(9): 968-978. |
| [11] | 苏琪辉, 王婧, 雒荣荣, 黄玉荣, 李欣, 王颖莉, 贾颖. 基于代谢组学与网络药理学研究四黑方对斑马鱼黑色素的作用机制[J]. 中国临床药理学与治疗学, 2024, 29(9): 988-1001. |
| [12] | 王丹, 张文艳, 罗仁洁, 陈元静, 韩雪, 曲波, 冯石芳, 聂夏子, 刘会玲. 分子对接在筛选抗宫颈癌药物中的研究进展[J]. 中国临床药理学与治疗学, 2024, 29(8): 955-960. |
| [13] | 李雯, 蒋虎刚, 王新强, 李应东, 刘凯, 赵信科. 基于网络药理学和实验验证探讨当归黄芪超滤物治疗放射性心肌纤维化的作用机制[J]. 中国临床药理学与治疗学, 2024, 29(6): 601-611. |
| [14] | 苏欣, 罗琳. UPLC-QE-MS联合网络药理学分析补中益气丸治疗溃疡性结肠炎的机制及实验验证[J]. 中国临床药理学与治疗学, 2024, 29(3): 241-251. |
| [15] | 张涛, 王若薇, 伏家林, 高悦, 胡名媛, 方正美, 陈燕, 姚应水. 基于网络药理学和体外实验探究α-亚麻酸改善神经炎症作用及机制[J]. 中国临床药理学与治疗学, 2024, 29(10): 1110-1119. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||