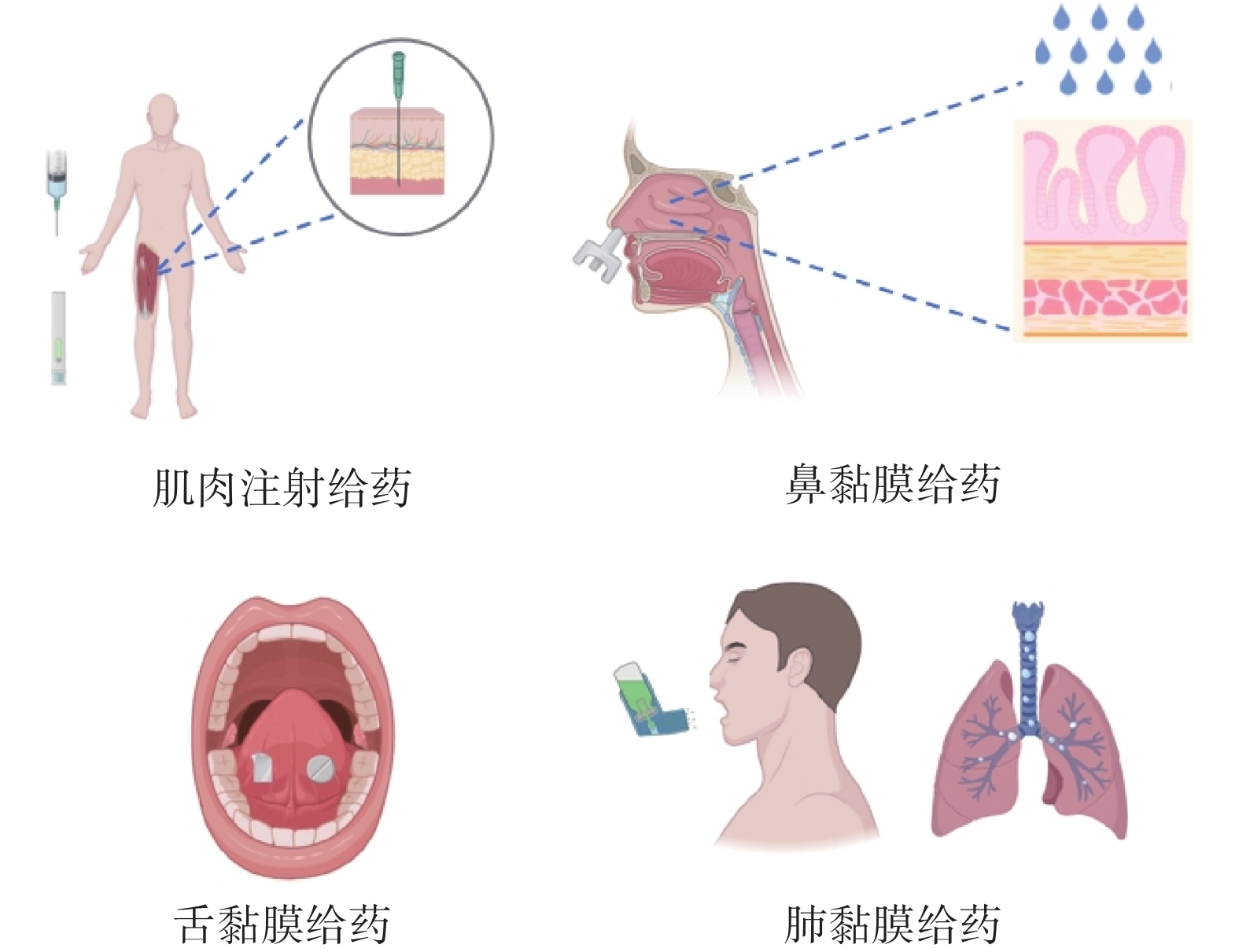
| 1 |
Khodoun MV, Strait RT, Hall A, et al. Importance of mast cell histamine secretion in IgG-mediated systemic anaphylaxis[J]. J Allergy Clin Immun, 2025, 155 (3): 956- 973.
doi: 10.1016/j.jaci.2024.11.009
|
| 2 |
LoVerde D, Iweala OI, Eginli A, et al. Anaphylaxis[J]. Chest, 2018, 153 (2): 528- 543.
doi: 10.1016/j.chest.2017.07.033
|
| 3 |
Dribin TE, Muraro A, Camargo Jr CA, et al. Anaphylaxis definition, overview, and clinical support tool: 2024 consensus report [J]. J Allergy Clin Immun, 2025. DOI: 10.1016/j.jaci.2025.01.021.
|
| 4 |
Fudge D, Viswesvaraiah G, John N. Anaphylaxis: a distributive emergency[J]. Medicine, 2025, 55 (3): 132- 137.
doi: 10.1016/j.mpmed.2020.12.006
|
| 5 |
Perlman L, Gabrielli S, Clarke AE, et al. Management of anaphylaxis after pre-hospital epinephrine use in children with food-induced anaphylaxis[J]. Ann Allerg Asthma Im, 2024, 133 (6): 682- 688.
doi: 10.1016/j.anai.2024.09.010
|
| 6 |
Simons FER, Simons KJ. Epinephrine (adrenaline) in anaphylaxis[J]. Chem Immunol Allergy, 2010, 95, 211- 222.
doi: 10.1159/000315954
|
| 7 |
Cardona V, Ansotegui IJ, Ebisawa M, et al. World allergy organization anaphylaxis guidance 2020[J]. World Allergy Organ, 2020, 13 (10): 100472.
doi: 10.1016/j.waojou.2020.100472
|
| 8 |
Muraro A, Roberts G, Worm M, et al. Anaphylaxis: guidelines from the European Academy of Allergy and Clinical Immunology[J]. Allergy, 2014, 69 (8): 1026- 1045.
doi: 10.1111/all.12437
|
| 9 |
李丽莎. 小儿严重过敏反应的早期识别与鉴别[J]. 中华临床免疫和变态反应杂志, 2021, 15 (4): 476- 477.
|
| 10 |
乔立俊. 浅析医疗机构制剂剂型的筛选[J]. 中国医疗前沿, 2007, 2 (21): 64.
|
| 11 |
Fang L, Wang L, Yao Y, et al. Micro-and nano-carrier systems: The non-invasive and painless local administration strategies for disease therapy in mucosal tissues[J]. Nanomed-Nanotechnol, 2017, 13 (1): 153- 171.
doi: 10.1016/j.nano.2016.08.025
|
| 12 |
Muraro A, Roberts G, Clark A, 等. 儿童严重过敏反应的处理: 欧洲变态反应学及临床免疫学会指南[J]. 中华临床免疫和变态反应杂志, 2009, 3 (1): 68- 77.
|
| 13 |
Simons FER, Gu X, Simons KJ. Epinephrine absorption in adults: Intramuscular versus subcutaneous injection[J]. J Allergy Clin Immun, 2001, 108 (5): 871- 873.
|
| 14 |
Basta C, Kumar V, Vastardi MA. Assessing epinephrine autoinjector use proficiency: patient and guardian practices at allergist visits[J]. J Allergy Clin Immun, 2024, 153 (2): AB91.
doi: 10.1016/j.jaci.2023.11.304
|
| 15 |
Dribin TE, Waserman S, Turner PJ. Who needs epinephrine? Anaphylaxis, autoinjectors, and parachutes[J]. J Aller Cl Imm-Pract, 2023, 11 (4): 1036- 1046.
doi: 10.1016/j.jaip.2023.02.002
|
| 16 |
Roy A, Geetha RV, Magesh A, et al. Autoinjector–a smart device for emergency cum personal therapy[J]. Saudi Pharm J, 2021, 29 (10): 1205- 1215.
doi: 10.1016/j.jsps.2021.09.004
|
| 17 |
Casale TB, Ellis AK, Nowak-Wegrzyn A, et al. Pharmacokinetics/pharmacodynamics of epinephrine after single and repeat administration of neffy, EpiPen, and manual intramuscular injection[J]. J Allergy Clin Immun, 2023, 152 (6): 1587- 1596.
doi: 10.1016/j.jaci.2023.08.007
|
| 18 |
杜鑫田. 严重过敏患者的“救命神器”——肾上腺素笔[J]. 健康, 2024, 45 (5): 70- 71.
|
| 19 |
Kessler C, Edwards E, Dissinger E, et al. Usability and preference of epinephrine auto-injectors: auvi-Q and EpiPen Jr[J]. Ann Allerg Asthma Im, 2019, 123 (3): 256- 262.
doi: 10.1016/j.anai.2019.06.005
|
| 20 |
Portnoy J, Wade RL, Kessler C. Patient carrying time, confidence, and training with epinephrine autoinjectors: the RACE survey[J]. J Aller Cl Imm-Pract, 2019, 7 (7): 2252- 2261.
doi: 10.1016/j.jaip.2019.03.021
|
| 21 |
Warren CM, Zaslavsky JM, Kan K, et al. Epinephrine auto-injector carriage and use practices among US children, adolescents, and adults[J]. Ann Allerg Asthma Im, 2018, 121 (4): 479- 489.
doi: 10.1016/j.anai.2018.06.010
|
| 22 |
Wood RA, Camargo Jr CA, Lieberman P, et al. Anaphylaxis in America: the prevalence and characteristics of anaphylaxis in the United States[J]. J Allergy Clin Immun, 2014, 133 (2): 461- 467.
doi: 10.1016/j.jaci.2013.08.016
|
| 23 |
Macadam C, Barnett J, Roberts G, et al. What factors affect the carriage of epinephrine auto-injectors by teenagers?[J]. Clin Transl Allergy, 2012, 2 (1): 3.
doi: 10.1186/2045-7022-2-3
|
| 24 |
Boswell B, Rudders SA, Brown JC. Emerging therapies in anaphylaxis: alternatives to intramuscular administration of epinephrine[J]. Curr Allergy Asthm R, 2021, 21 (3): 18.
doi: 10.1007/s11882-021-00994-0
|
| 25 |
Prince BT, Mikhail I, Stukus DR. Underuse of epinephrine for the treatment of anaphylaxis: missed opportunities[J]. J Asthma Allergy, 2018, 11, 143- 151.
doi: 10.2147/JAA.S159400
|
| 26 |
Fromer L. Prevention of anaphylaxis: the role of the epinephrine auto-injector[J]. Am J Med, 2016, 129 (12): 1244- 1250.
doi: 10.1016/j.amjmed.2016.07.018
|
| 27 |
Vitore JG, Bharathi K, Salave S, et al. Intranasal transmucosal drug delivery: an alternative approach to the parenteral route for medical emergencies[J]. J Drug Deliv Sci Tec, 2023, 83, 104421.
doi: 10.1016/j.jddst.2023.104421
|
| 28 |
Ebisawa M, Lowenthal R, Tanimoto S, et al. Neffy, epinephrine nasal spray, demonstrates a positive efficacy and safety profile for the treatment of allergic reactions in pediatric patients at-risk of anaphylaxis: phase 3 study results[J]. J Allergy Clin Immun, 2024, 153 (2): AB371.
doi: 10.1016/j.jaci.2023.11.888
|
| 29 |
Oppenheimer J, Casale TB, Camargo CA, et al. Upper respiratory tract infections have minimal impact on neffy's pharmacokinetics or pharmacodynamics[J]. J Aller Cl Imm-Pract, 2024, 12 (6): 1640- 1643.
doi: 10.1016/j.jaip.2024.02.038
|
| 30 |
Van Kempen M, Bachert C, Van Cauwenberge P. An update on the pathophysiology of rhinovirus upper respiratory tract infections[J]. Rhinology, 1999, 37 (3): 97- 103.
doi: 10.7248/jjrhi1982.38.4_389
|
| 31 |
Lapidot T, Tal Y, Megiddo D, et al. Fast acting, 5 years stable intra-nasal powder-spray epinephrine for anaphylaxis: Integrated analysis of two clinical trials[J]. J Allergy Clin Immun, 2025, 155 (2): AB94.
doi: 10.1016/j.jaci.2024.12.298
|
| 32 |
Tal Y, Ribak Y, Rubin L, et al. Fast acting, dry powder, needle-free, Intranasal epinephrine spray: a promising future treatment for anaphylaxis[J]. J Aller Cl Imm-Pract, 2023, 11 (10): 3047- 3054.
doi: 10.1016/j.jaip.2023.06.044
|
| 33 |
Fasiolo LT, Manniello MD, Tratta E, et al. Opportunity and challenges of nasal powders: drug formulation and delivery[J]. Eur J Pharm Sci, 2018, 113, 2- 17.
doi: 10.1016/j.ejps.2017.09.027
|
| 34 |
Djupesland PG. Nasal drug delivery devices: characteristics and performance in a clinical perspective-a review[J]. Drug Deliv Transl Re, 2013, 3 (1): 42- 62.
doi: 10.1007/s13346-012-0108-9
|
| 35 |
Djupesland PG, Skretting A. Nasal deposition and clearance in man: comparison of a bidirectional powder device and a traditional liquid spray pump[J]. J Aerosol Med Pulm D, 2012, 25 (5): 280- 289.
doi: 10.1089/jamp.2011.0924
|
| 36 |
Lapidot T, Bouhajib M, Faulknor J, et al. A novel faster-acting dry powder-based, naloxone intranasal formulation for opioid overdose[J]. Pharm Res-Dordr, 2022, 39 (5): 963- 975.
doi: 10.1007/s11095-022-03247-5
|
| 37 |
Adler JT, Meyer-Rochow GY, Chen H, et al. Pheochromocytoma: Current approaches and future directions[J]. Oncologist, 2008, 13 (7): 779- 793.
doi: 10.1634/theoncologist.2008-0043
|
| 38 |
Lee B, Gauld R, Kim H. The risk of severe adverse reactions to neffy intranasal epinephrine spray[J]. J Allergy Clin Immun, 2024, 153 (2): 535- 536.
doi: 10.1016/j.jaci.2023.11.015
|
| 39 |
Roma JR, Rebollo PC, Bastida C. Sublingual and buccal drug administration in medical emergencies[J]. Med Clin-Barcelona, 2024, 163 (12): 619- 625.
|
| 40 |
Rawas-Qalaji MM, Simons FER, Simons KJ. Sublingual epinephrine tablets versus intramuscular injection of epinephrine: dose equivalence for potential treatment of anaphylaxis[J]. J Allergy Clin Immun, 2006, 117 (2): 398- 403.
doi: 10.1016/j.jaci.2005.12.1310
|
| 41 |
Birudaraj R, Berner B, Shen S, et al. Buccal permeation of buspirone: mechanistic studies on transport pathways[J]. J Pharm Sci-Us, 2005, 94 (1): 70- 78.
doi: 10.1002/jps.20208
|
| 42 |
Rachid O, Rawas-Qalaji MM, Simons FER, et al, Epinephrine(adrenaline) absorption from new-generation, taste-masked sublingual tablets: a preclinical study [J]. J Allergy Clin Immun, 2013, 131(1): 236-238.
|
| 43 |
Rawas-Qalaii M, Bafail R, Cagliani R, et al. Assessment of epinephrine sublingual stability and permeability pathways to enhance its permeability for the treatment of anaphylaxis[J]. Eur J Pharm Sci, 2021, 167, 106025.
doi: 10.1016/j.ejps.2021.106025
|
| 44 |
Rawas-Qalaji M, Alozi M, Khan SA, et al. Sublingual route for the continuous release of epinephrine from chitosan nanoparticles[J]. J Drug Deliv Sci Tec, 2024, 101, 106246.
doi: 10.1016/j.jddst.2024.106246
|
| 45 |
Liu AS, Nargozian CD, Greene AK. Subcutaneous epinephrine for vasoconstriction: an evidence-based evaluation[J]. Plast Reconstr Surg, 2010, 126 (3): 157- 158.
doi: 10.1097/PRS.0b013e3181e3b5c1
|
| 46 |
Freedman M, Oppenheimer J, Wargacki S, et al. Pharmacokinetics study of epinephrine sublingual film: results form the formulation and dosage selection[J]. Ann Allerg Asthma Im, 2022, 129 (5): S10.
doi: 10.1016/j.anai.2022.08.533
|
| 47 |
Kraus CN, Wargacki S, Golden D, et al. Integrated phase I pharmacokinetics and pharmacodynamics of epinephrine administered through sublingual film, autoinjector, or manual injection [J]. Ann Allerg Asthma Im, 2025. DOI: 10.1016/j.anai.2025.01.006.
|
| 48 |
Oppenheimer J, Golden D, Camargo C, et al. Impact of food exposure on the pharmacokinetics of epinephrine sublingual film[J]. J Allergy Clin Immun, 2023, 151 (2): AB2.
doi: 10.1016/j.jaci.2022.12.013
|
| 49 |
Wargacki S, Slatko G, Haney M, et al. Impact of angioedema on the pharmcokinetics of epinephrine following administration via sublingual film[J]. Ann Allerg Asthma Im, 2023, 131 (5): S16.
doi: 10.1016/j.anai.2023.08.057
|
| 50 |
Rau JL. The inhalation of drugs: advantages and problems[J]. Resp Care, 2005, 50 (3): 367- 382.
|
| 51 |
Kerwin EM, Marrs T, Luo MZ, et al. Pharmacokinetic study of epinephrine hydrofluoroalkane (Primatene MST) metered-dose inhaler[J]. J Aerosol Med Pulm D, 2020, 33 (5): 282- 287.
doi: 10.1089/jamp.2019.1577
|
| 52 |
Schlegel C, Fux R, Biedermann T. Epinephrine inhalers in emergency sets of patients with anaphylaxis[J]. J Dtsch Dermatol Ges, 2009, 7 (5): 420- 425.
doi: 10.1111/j.1610-0387.2008.06938.x
|
| 53 |
Simons FER, Gu X, Johnston LM, et al. Can epinephrine inhalations be substituted for epinephrine injection in children at risk for systemic anaphylaxis?[J]. Pediatrics, 2000, 106 (5): 1040- 1044.
doi: 10.1542/peds.106.5.1040
|
| 54 |
张郃, 李硕, 程艺, 等. 无针注射技术的研究进展[J]. 药学学报, 2024, 59 (3): 591- 599.
doi: 10.16438/j.0513-4870.2023-0853
|