| 1 |
Lal S. Primary Sjögren's syndrome[J]. N Engl J Med, 2018, 379 (1): 96- 97.
doi: 10.1056/NEJMc1804598
|
| 2 |
Qin B, Wang J, Yang Z, et al. Epidemiology of primary Sjogren's syndrome: a systematic review and meta-analysis[J]. Ann Rheum Dis, 2015, 74 (11): 1983- 1989.
doi: 10.1136/annrheumdis-2014-205375
|
| 3 |
Zhang Q, Lv X, Wang Y, et al. Expression of CFTR, a hallmark gene of ionocytes, is downregulated in salivary glands of Sjögren's syndrome patients[J]. Arthritis Res Ther, 2022, 24 (1): 263.
doi: 10.1186/s13075-022-02959-8
|
| 4 |
Bjordal O, Norheim KB, Rødahl E, et al. Primary Sjögren's syndrome and the eye[J]. Surv Ophthalmol, 2020, 65 (2): 119- 132.
doi: 10.1016/j.survophthal.2019.10.004
|
| 5 |
Horeth E, Oyelakin A, Song EC, et al. Transcriptomic and single-cell analysis reveals regulatory networks and cellular heterogeneity in mouse primary Sjögren's syndrome salivary glands[J]. Front Immunol, 2021, 12, 729040.
doi: 10.3389/fimmu.2021.729040
|
| 6 |
Kroese FGM, Haacke EA, Bombardieri M. The role of salivary gland histopathology in primary Sjögren's syndrome: promises and pitfalls[J]. Clin Exp Rheumatol, 2018, 112 (3): 222- 233.
|
| 7 |
Jonckheere S, Adams J, de Groote D, et al. Epithelial-mesenchymal transition (EMT) as a therapeutic target[J]. Cells Tissues Organs, 2021, 5, 1- 26.
doi: 10.3389/978-2-83251-634-8
|
| 8 |
邹芳霞, 王钢, 漆文霞. 基于“肺与大肠相表里”理论探讨滋阴润燥丸治疗原发性SS[J]. 中医药临床杂志, 2022, 34 (2): 211- 214.
|
| 9 |
Roszkowska AM, Rauz S, Szczepańska A, et al. Ophthalmologic manifestations of primary Sjögren's syndrome[J]. Genes (Basel), 2021, 12 (4): 542.
|
| 10 |
Singh N, Cohen PL. The T cell in sjogren's syndrome: Force majeure, not spectateur[J]. Autoimmun, 2012, 39 (3): 229- 233.
doi: 10.1016/j.jaut.2012.05.019
|
| 11 |
Zhao H, Li Q, Ye M, et al. Tear luminex analysis in dry eye patients[J]. Med Sci Monit, 2018, 24, 7595- 7602.
doi: 10.12659/msm.912010
|
| 12 |
Chen X, Aqrawi LA, Utheim TP, et al. Elevated cytokine levelsin tears and saliva of patients with primary Sjögren's syndrome correlate with clinical ocular and oral manifestations[J]. Sci Rep, 2019, 9, 7319.
doi: 10.1038/s41598-019-43714-5
|
| 13 |
Yin H, Vosters JL, Roescher N, et al. Location of immunization and interferon-gamma are central to induction of salivary gland dysfunction in Ro60 peptide immunized model of Sjogren's syndrome[J]. PLoS One, 2011, 6, e18003.
doi: 10.1371/journal.pone.0018003
|
| 14 |
Torres-Aguilar H, Sosa-Luis SA, Aguilar-Ruiz SR. Infections as triggers of flares in systemic autoimmune diseases: Novel innate immunity mechanisms[J]. Curr Opin Rheumatol, 2019, 31, 525- 531.
doi: 10.1097/BOR.0000000000000630
|
| 15 |
Yoshimura S, Nakamura H, Horai Y, et al. Abnormal distribution of AQP5in labial salivary glands is associated with poor saliva secretion in patients with Sjögren's syndrome including neuromyelitis optica complicated patients[J]. Mod Rheumatol, 2016, 26, 384- 390.
doi: 10.3109/14397595.2015.1083146
|
| 16 |
Caraba A, Iurciuc S, Nicolin M, et al. Endothelial dysfunction in primary Sjögren's syndrome: correlation with serum biomarkers of disease activity[J]. Int J Mol Sci, 2023, 24 (18): 13918.
doi: 10.3390/ijms241813918
|
| 17 |
Abe S, Tsuboi H, Kudo H, et al. M3 muscarinic acetylcholine receptor-reactive Th17 cells in primary Sjögren's syndrome[J]. JCI Insight, 2020, 5 (15): e135982.
|
| 18 |
Steinfeld S, Cogan E, King LS, et al. Abnormal distribution of aquaporin-5 water channel protein in salivary glands from Sjögren syndrome patients[J]. Lab Invest, 2001, 81 (2): 143- 148.
doi: 10.1038/labinvest.3780221
|
| 19 |
Ishibashi K, Kondo S, Hara S, et al. The evolutionary aspects of aquaporin family[J]. Am J Physiol Regul Integr Comp Physiol, 2011, 69 (3): R566- R576.
doi: 10.1007/978-94-024-1057-0_2
|
| 20 |
Tzartos JS, Stergiou C, Daoussis D, et al. Antibodies to aquaporins are frequentin patients with primary Sjögren's syndrome[J]. Rheumatology (Oxford), 2017, 56 (12): 2114- 2122.
doi: 10.1093/rheumatology/kex328
|
| 21 |
Konttinen YT, Tensing EK, Laine M, et al. Abnormal distribution of aquaporin-5 in salivary glands in the NOD mouse model for Sjögrens syndrome[J]. J Rheumatol, 2005, 32 (6): 1071- 1075.
|
| 22 |
李晓宇, 韩星海. SS与水通道蛋白-5[J]. 中华风湿病学杂志, 2017, 11 (6): 378- 380.
|
| 23 |
阮光峰. 水通道蛋白5在SS发病机制中的作用研究进展[J]. 同济大学学报: 医学版, 2016, 37 (2): 113- 117.
|
| 24 |
Lee SM, Lee SW, Kang M, et al. FoxO1 as a regulator of aquaporin 5 expression in the salivary gland[J]. Dent Res, 2021, 100 (11): 1281- 1288.
doi: 10.1177/00220345211003490
|
| 25 |
Alam J, Koh JH, Kwok SK, et al. Functional epitopes for anti-aquaporin 5 antibodies in Sjögren syndrome[J]. J Rheumatol, 2017, 44 (9): 1317- 1325.
doi: 10.1177/0022034517717965
|
| 26 |
Shaalan A, Carpenter G, Proctor G. Epithelial disruptions, but not immune cell invasion, induced secretory dysfunction following innate immune activation in a novel model of acute salivary gland injury[J]. Oral Pathol Med, 2018, 47 (2): 211- 219.
doi: 10.1111/jop.12663
|
| 27 |
Nguyen CQ, Peck AB. The interferon-signature of Sjögren's syndrome: How unique biomarkers can identify underlying inflammatory and immunopathological mechanisms of specific diseases[J]. Front Immunol, 2013, 4, 142.
doi: 10.3389/fimmu.2013.00142
|
| 28 |
Mona M, Mondello S, Hyon JY, et al. Clinical usefulness of anti-muscarinic type 3 receptor autoantibodies in patients with primary Sjögren's syndrome[J]. Clin Exp Rheumatol, 2021, 39 (4): 795- 803.
doi: 10.55563/clinexprheumatol/gy6udz
|
| 29 |
Choi SS, Jang E, Jang K, et al. Autoantibody-mediated dysfunction of salivaryglands leads to xerostomia in SKG mice[J]. Immune Network, 2019, 19 (6): e44.
doi: 10.4110/in.2019.19.e44
|
| 30 |
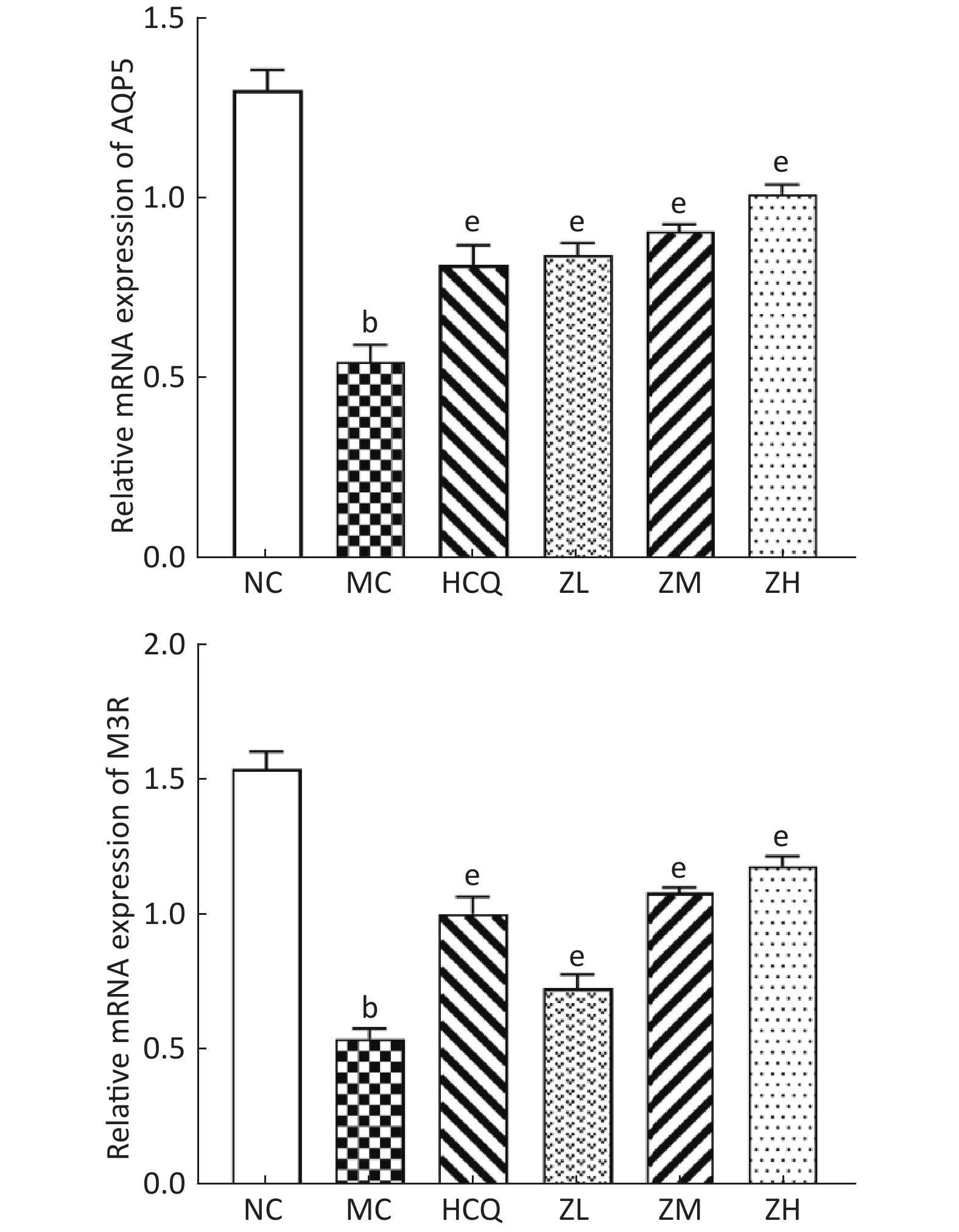
秦源, 林崇泽, 孙晗, 等. 滋阴润燥丸对SS颌下腺细胞AQP5及M3R表达的影响[J]. 中国中医急症, 2017, 26 (1): 5- 8.
|